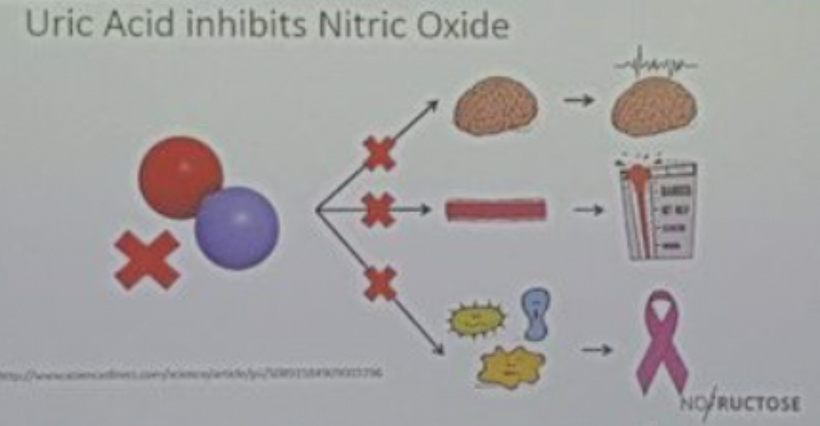
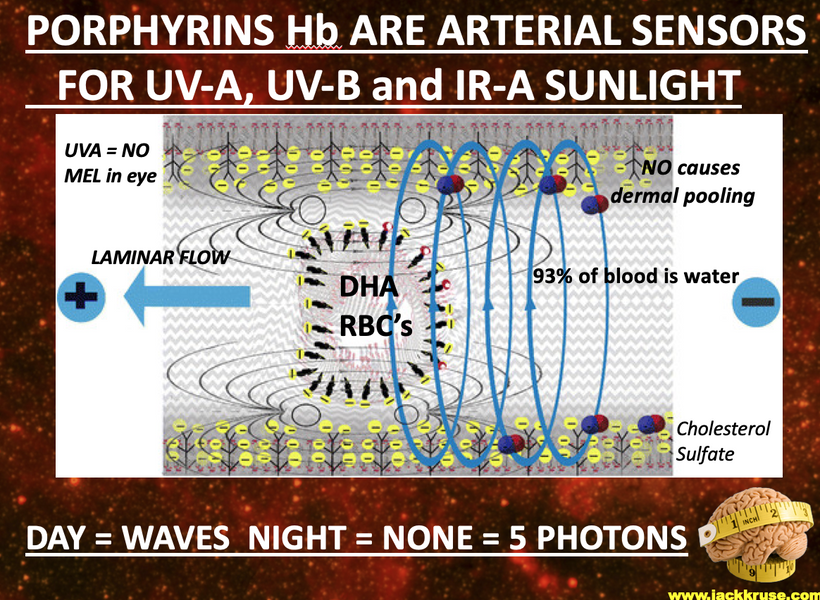
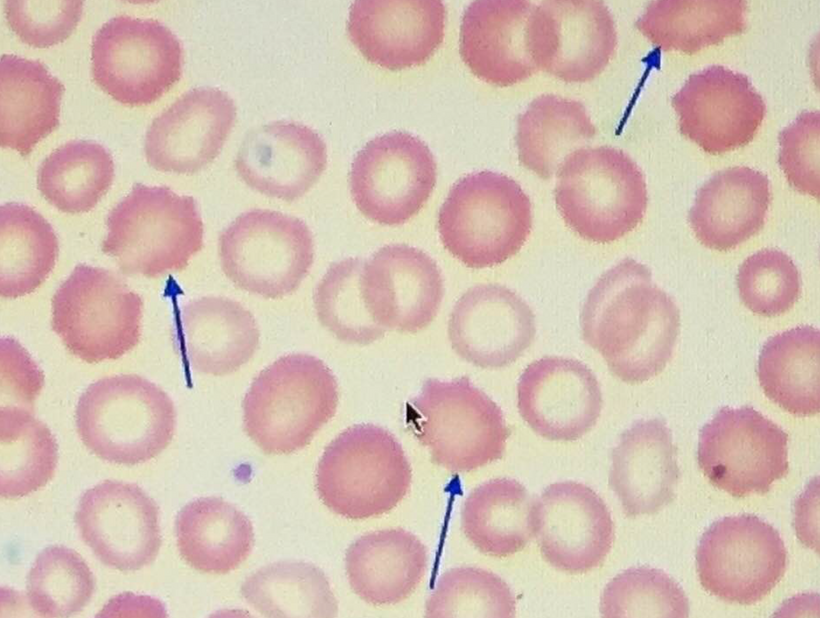
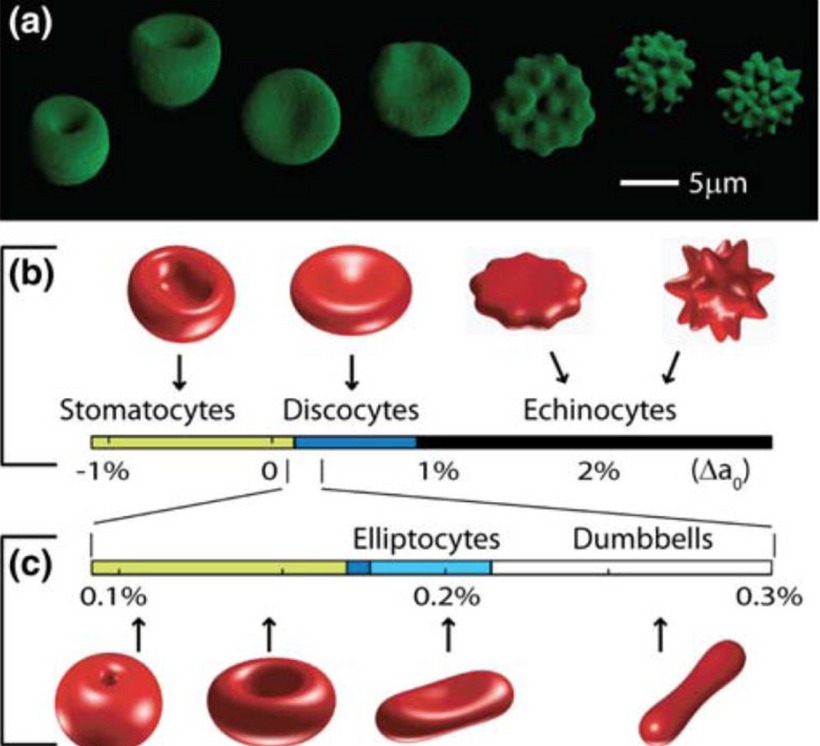
Sneezing in bright sunlight, often referred to as the photic sneeze reflex or “sun sneezing,” is a fascinating phenomenon that involves an intricate interplay between the optic nerve and the olfactory system in the brain. Let me break it down based on the concepts I’ve introduced you to in this series.
The reflex likely stems from a surge of high-intensity light stimulating the optic nerve, which carries electrical signals from the retina in the eye to various brain regions. The sensory cranial nerve that innervates the eye and nose is the trigeminal nerve. The ophthalmic division innervates the eye and the maxillary division innervates the nose. Centralized biology believes currently that the ophthalmic (CN V1) and maxillary (CN V2) divisions of the trigeminal nerve (CN V) do not have a tight phylogenetic coupling with the olfactory nerve (CN I) and optic nerve (CN II). The reason they believe this is because they do not yet understand how UPEs operate in the CNS and PNS of man. This is why they believe these cranial have distinct evolutionary origins and functions. The decentrlaized viewpoint is quite different. This blog explores why humans still have the photic sneeze reflex, also known as the ACHOO syndrome . ACHOO stands for Autosomal dominant compleling helio-opthalmic outburst syndrome. It’s an inherited autosomal dominant reflex that causes people to sneeze after sudden exposure to bright light, like moving from a dim room into sunlight.
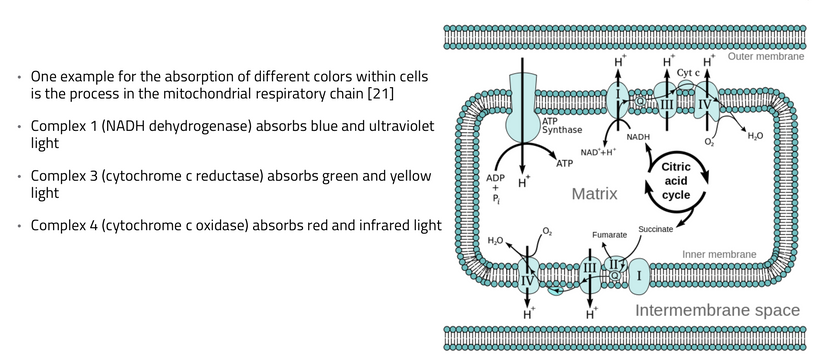
The retina contains retinal ganglion cells, whose axons form the optic nerve. In areas like the fovea, these cells are highly sensitive due to their connection to fewer photoreceptors, making them particularly responsive to bright light. As light enters the eye, it triggers an action potential, a rapid electrical impulse, along the optic nerve, accompanied by the release of water as part of the neuronal firing process. As a result, UPEs are created within the nerves in reponse to light. The UPE created is stochastically related to myelination level, mitochondrial density, and heteroplasmy rate of the neural tract in question.
KEY DECENTRALIZED POINT: UPEs are the basis of indirect neural wiring of the CNS and PNS in eukaryotes. This is a big missing piece in centralized biology now. Visual information from the optic nerve (CN II) can be indirectly relayed to the olfactory system (CN I) and the trigeminal system (CN V) via multisensory integration pathways in the brain, particularly involving structures like the hypothalamus, the limbic system, and regions of the ventral prefrontal cortex. If melanin in any are is missing or destroyed this would affect the resultant UPE. The same is true with mitochondrial heteroplasmy or with myelination status.
This neural optical signal travels through the optic nerve, passing through the optic chiasm (where some fibers cross to the opposite side of the brain) and onward to structures like the lateral geniculate nucleus and pretectal area. However, the key interaction for the photic sneeze reflex seems to occur in the midbrain and forebrain, where the optic nerve’s activity can influence nearby neural pathways, including those tied to the olfactory bulb.
The cerebral cortex can be divided into three regions based on phylogeny. The archicortex is the most primitive domain and consists of the olfactory bulbs, olfactory tracts, and olfactory cortex (the piriform lobe of mammals). The paleocortex, the next oldest cortical region, is the cortical seat of the limbic system. The neocortex is the newest real estate from an evolutionary standpoint, and it consists of the frontal (rostral), parietal (dorsolateral), temporal (ventrolateral), and occipital (caudal) lobes. Interspecies differences in cortical function hinge on the ratio of paleocortex (emotion-driven and involuntary functions) to that of neocortex (higher associative and cognitive function).

The olfactory bulb, an evolutionarily older part of the brain with a three-layered paleocortex (unlike the six-layered neocortex of the visual system), is less myelinated and has a lowered mitochondrial density and this makes it more vulnerable to overstimulation. I’ve mentioned melanosomes in this region which are pigment-containing structures in the olfactory bulb, which play a role in modulating this response. A deficiency in melanosomes or myelin, change the electrical resistance in the region. This can be linked to reduced melanin production from proopiomelanocortin (POMC), dehydration of melanin in this area, or due to a lack of myelination in this are leaves the olfactory bulb hypersensitive to acute overwhelming stimuli. When the optic nerve fires intensely due to bright light, this electrical surge “spills over” into the olfactory pathways, triggering a reflexive sneeze.
TRIGEMINAL NERVE = CN V
This nerve has 3 divisions and it innervates the eye and nose for sensation. This information is indirectly shared with the optic and olfactory nerve. The trigeminal ganglion, from which the nerve emerges, does not have layers of cortex associated with it. It is a peripheral sensory ganglion, analogous to the dorsal root ganglia of the spinal cord, and consists of clusters of sensory neuron cell bodies surrounded by satellite glial cells and enclosed in a dural capsule. You can see the ganglion below as the large yellow structure on the left that is giving three divisions called the ganglion Trigeminale. The top trunk goes to the eye (opthalamic), the middle trunk goes to the nose (maxillary), and the bottom trunk goes to the mouth and jaw (mandibular).

Trigeminal Ganglion (CN V): This is a collection of neuron cell bodies outside the central nervous system (CNS). Sensory information from the trigeminal nerve is transmitted from the ganglion to the trigeminal nuclei in the brainstem, which then relay the information to the thalamus and finally to the somatosensory cortex of the brain. The cortex itself (the outer layer of the brain) has six cellular layers, but these are part of the brain, not the ganglion.
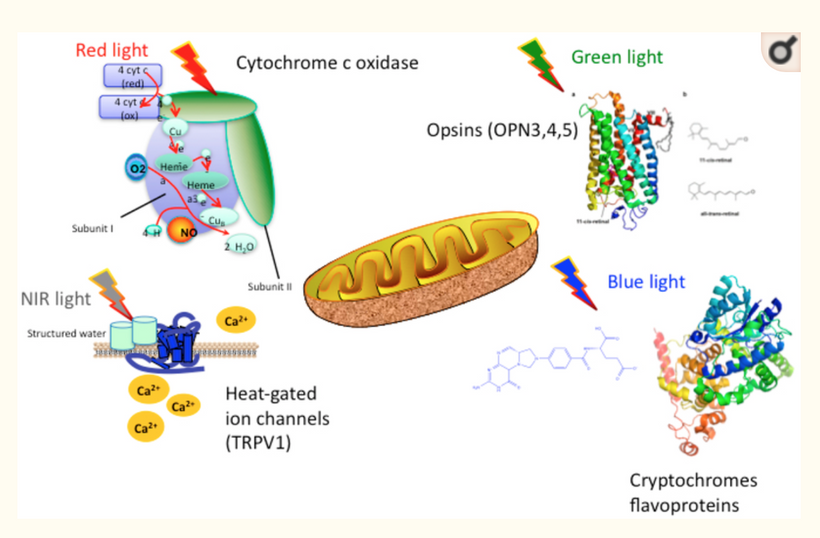
A similar mechanism is likely playing a role in part of the brain sensitive to photosensitive seizures in some people who have evidence of blue light hazard in the neocortex. This is more common in people with ASD, OCD, ADHD, and mental disorders linked to the frontal lobes and medial temporal lobes. Since the brain accounts for 20% of the human body’s energy usage, when it encounters a change in electrical resistance this sets up a potential problem. This can be from a transgeneration alteration of brain topograghy or it can happen from the blue light hazard, or spike protein damage, that destroys mitochondrial density and myelin density in adjacent neural structures leading to pathology like spikes of seizures on the brain.
OLFACTORY BULB ANATOMY
The three-layered cortex, including the olfactory bulb, does have myelin, but its distribution and density differ from the six-layered neocortex rregions in humans. Myelin, produced by oligodendrocytes in the central nervous system, insulates axons to speed up electrical signal transmission.
Within the Olfactory Bulb: The bulb itself contains a mix of myelinated and unmyelinated axons. The olfactory nerve (cranial nerve I), which carries signals from the nasal cavity to the bulb, is unique because its axons are largely unmyelinated—these are ensheathed by olfactory ensheathing cells (a type of glia) rather than traditional myelin from Schwann cells or oligodendrocytes. However, once inside the olfactory bulb, some of the deeper axons, especially those from mitral and tufted cells that project outward to other brain regions (e.g., via the olfactory tract), are myelinated. The bulb’s internal circuitry, like the connections between granule cells and mitral cells, includes a mix of both types, but myelination is less extensive than in neocortical areas.
Olfactory Tract and Beyond: As signals leave the olfactory bulb via the olfactory tract toward areas like the piriform cortex or amygdala, myelination increases. These projection axons are more consistently myelinated to support faster transmission over longer distances.
In other three-layered regions, like the hippocampus (archicortex), myelination is present but varies. For example, the mossy fibers in the hippocampus are famously unmyelinated, while other output pathways, like the Schaffer collaterals, have myelin.
OTHER PALEOCORTEX SITES IN HUMANS
Hippocampal Formation (Part of the Archicortex)
The hippocampus and its associated structures, like the dentate gyrus, are often classified as archicortex, a subtype of three-layered cortex. Located within the medial temporal lobe, these areas are part of the limbic system and play a major role in memory formation and spatial navigation. While the hippocampus itself has a unique cellular organization (e.g., the CA fields), its cortical structure is simpler than the neocortex and aligns with the older, three-layered design. The hippocampus is critical in learning and memory in humans.
Parahippocampal Gyrus (Transitional Zone)
Parts of the parahippocampal gyrus, which surrounds the hippocampus, contain transitional zones between the three-layered paleocortex/archicortex and the six-layered neocortex. The entorhinal cortex, for instance, acts as a gateway between the hippocampus and neocortex and has a mix of layering, but its deeper roots tie to the older cortical structure. The parahippocampus plays a crucial role in memory and navigation, primarily by processing contextual associations.
Its functions includeepisodic memory encoding (especially for context), visuospatial processing and scene recognition (via the parahippocampal area, or PPA), and binding objects with their surroundings.
Cingulate Gyrus (Partial Overlap)
The cingulate cortex, part of the limbic system above the corpus callosum, has regions that transition from three-layered to six-layered cortex. The more anterior and deeper parts are closer to the paleocortical structure and are involved in emotion and autonomic regulation. This is more sensitive to damage in the neural and vascular networks because of the changes in electrical resistance.
These changes in the neural networks have different electrical resistances, as such, acutre high duration stimuli cause aberrent signaling and these interactions are exacerbated by factors like blue light or non-native electromagnetic frequency (nnEMF) exposure, which disrupt melanin, dopamine, myelin and melatonin levels in the forebrain.
A deficiency in any of these neurochemicals will weaken the brain’s ability to dampen this cross-talk due to the change in electrical resistance, making the reflex more pronounced at those times. This is why defects in this area are associated with OCD and ADHD. This is also why EHS includes these diagnosises under their umbrella. The cingulate gyrus is a key part of the limbic system that helps regulate emotions, emotional responses to pain, and social behavior. It also plays a role in learning, memory, attention, decision-making, and coordinating sensory input with emotional and motor responses.
In cases where sneezing occurs even in low light, it could indicate a severe imbalance, of melanin, myeling or represent an excessive conversion of melanin to dopamine (L-DOPA), further depleting forebrain reserves and leading to mental disease in the limbic circuits. It is clue to the decentralized clinician that something is amiss in this region.
Is Myelin Deficient in These Regions Like Mitochondrial Density?
Yes, myelin tends to be less dense or less uniformly distributed in three-layered cortex regions compared to the neocortex, and this parallels the lower mitochondrial density in the paleocortex. This is why so many neurodegenerative disorders have memory loss as one of the first features because of this hybrid design.
Olfactory Bulb: The function of the olfactory bulb is to process smell information by receiving signals from the nose and relaying them to other parts of the brain for interpretation. It acts as the first processing center for smells, organizing odor data through a spatial map in its glomeruli layer and transmitting this information to areas involved in perception, emotion, and memory. It mimics the function of the retina as a processor for light signals.
The olfactory bulb’s simpler three-layered structure and its role as a direct sensory processor mean it relies less on heavily myelinated, high-speed pathways within its local circuits. The lower myelin presence aligns with its evolutionary age and lower mitochondrial density. Fewer mitochondria mean less energy available to support the production and maintenance of myelin, which is an energy-intensive process. This makes the bulb more vulnerable to overstimulation or mitochondrial damage. This is why strong light can overwhelm the wiring diagram and mitochondria leading to aberrent UPE release in the bulb that results in the photic sneeze reflex. Less myelin = less stability to environental triggers = more sensitive to the environment because myelin helps stabilize and regulate neuronal firing.
Hippocampus: Similarly, the hippocampus has patchy myelination. Its older architecture and lower mitochondrial density (compared to neocortical areas like the prefrontal cortex) suggest a reduced capacity for energy-hungry processes like extensive myelination. This contributes to its sensitivity to stressors or metabolic imbalances. Patchy myelination = a more sensitive regions to learn about environmental changes for the animal.
The hippocampus is crucial in humans for forming new memories, especially declarative (episodic and semantic) memories, and for spatial navigation. It converts short-term memories into long-term ones, helps with the retrieval of memories, and plays a role in emotional responses and regulating other brain functions.
Comparison to Neocortex: The six-layered neocortex, especially in areas like the visual or motor cortex, has a much higher density of myelinated axons, supported by greater mitochondrial populations to meet the energy demands of complex, rapid processing. The neocortex’s white matter tracts, like the corpus callosum, are heavily myelinated, reflecting its evolutionary specialization for speed and efficiency over environmental processing of information data collection.

The parallel between lower mitochondrial density and reduced myelination in three-layered cortex makes sense biologically from the evolutionary lens. Mitochondria provide the ATP needed for myelin synthesis and maintenance, so a region with fewer mitochondria might naturally have less myelin and this make . This would leave areas like the olfactory bulb more prone to “cross-talk” or overstimulation—like the optic nerve’s surge triggering a sneeze—since myelin normally dampens excessive signal spread. A deficiency in melanosomes or neurochemicals (e.g., dopamine, melatonin) could exacerbate this by further stressing the system’s energy balance.
So, while these regions aren’t completely devoid of myelin, they’re relatively deficient compared to evolutionary newer brain areas, much like their mitochondrial profiles. This appears to be a design feature of older cortex which favoring flexibility or redundancy over speed but it could also signal vulnerability under modern conditions like nnEMF or blue light exposure.
SUMMARY
From a decentralized medicine perspective, this reflex serves as a diagnostic clue to ongoing mitochondria damage due to some cause. Why? I believe this reflex used to be used to spread herd immunity in primitive humans and primates.
Chronic photic sneezing signal underlying issues—dopamine or melatonin deficiencies, myelin degradation, or even early neurodegeneration, potentially tied to modern environmental stressors like excessive screen time or artificial lighting. The link to the PNAS article referenced below (1988 Cite 5) suggests a deeper exploration of neural connectivity, possibly hinting at how melanopsin, a light-sensitive pigment in retinal ganglion cells ties into this broader system.
In short, sneezing in bright sunlight reflects a quirky thing today, but really is a beacon of a novel past use because of the indirect neural cross-wiring diagrams, where light overload in the optic nerve or trigeminal nerve tickles the olfactory system into a sneeze for some reason. I believe that reason was pathogens. Inside of caves where early primates and humans lived if one was sick from a viral or bacterial disease sneezing there would have infected the tribe with no way to limit the infection. If the sick pre human however, emerged from its cave to bright sunlight, this would have been the ideal time to expel a pathogen to expose the tribe to it, while the sun could disinfect and immunized the tribe to the pathogen more safely. It’s a reminder of how interconnected our sensory pathways are and how imbalances in one area might ripple across others and into the thalamus and out immune system. Many people have forgotten what I wrote in the brain gut series that the formation of the human brain and immune system are linked deeply phylogenetically.
We have specialized Immune Cells in the Brain: The brain contains resident innate immune cells, called microglia, which have a distinct developmental origin from peripheral immune cells but are crucial for normal brain development and function, further illustrating a specialized, co-evolved relationship.
Recent Discoveries: The relatively recent discovery of functional lymphatic vessels in the brain meninges (previously thought not to exist) has provided a physical “missing link” for direct communication between the central nervous system and the peripheral immune system, highlighting an anatomical connection that was likely maintained throughout evolution for some reason.
I believe, this is why many currentl viral diseases affect smell and taste function when we get infected. What do you think about this as a marker for broader health insights? Might it be a clue that your environment is not as good as you think? Might it be a signal to your family and friends that your sick and they need to be made aware of it in some way before we were able tp speak and communicate it?
I believe the ACHOO reflex is designed to not to afflicted all tribe members by design. Primates and humans are social so not everyone would need the ability to tell the tribe one was sick. I believe the ACHOO reflex was designed by nature to be caused epigenetically due to aberrent UPE signaling during brain and immune development in the primate tree.
I think it is a marker for an aberrent immunse system in modern humans. It is not a formal genetic disease, but a casual base pair change due to optical signaling in the brain. It is caused by one base change of a C to a T in an intergenic region of chromosome two. There is correlative evidence that every copy of this single nucleotide polymorphism (SNP) is associated with a 1.3x increase in likelihood of having the photic sneeze reflex. I believe the change is caused by altered UPE firing due to the unique characteristics of the olfactory bulb. In fact, today I believe this reflex is a remnant of phylogeny of how we handled communicable diseases before we had our modern human immune system. In other words, it was Nature’a original vaccine system to protect the herd before we could talk.
In centralized medicine the exact mechanism is not fully understood but decentralized medicine focus on light has a rather unique way of looking this disease. The small base pair change involve a crossed signal between the optic (vision) and trigeminal (sneeze) nerves. The opthalmic and maxillary division of the trigeminal nerve have a tight phylopgenic coupling and since this nerve innervates tissues with the environment this was a way to sense and expel pathogens from the nasal and repsiratory tree while in bright light so that other organisms could be infected with this pathogen at the same time they were in sunlight with UV and NIR light to confer infection and photorepair prior to our highly developed immune systems in the eukaryotic tree. So effectively, this reflex is a remnant of our past. This reflex can be triggered by sunlight, camera flashes, or artificial lights and affects up to 35% of the population. That is how I see it. How do you see it?
CITES
Mosharov, E. V. et al. Nature https://doi.org/10.1038/s41586-025-08740-6 (2025).
Padamsey, Z. & Rochefort, N. L. Curr. Opin Neurobiol. 78, 102668 (2023).
https://www.nature.com/articles/d41586-024-03716-4