Russell van Gelder, M.D., Ph.D., a professor of ophthalmology at the University of Washington, studied the circadian rhythms of genetically tweaked mice that were missing rod and cone cells and melanopsin. As expected, the circadian rhythms of these mice continued cycling but could no longer adapt to changes in light exposure. Surprisingly, though, the activity patterns of their retina’s were still responsive to light changes, suggesting that there was another light photoreceptor in play in the eye and skin. That pigment was neuropsin.
This opsin, like all opsins, work with the Vitamin A cycle in the body. Most people struggle to convert dietary carotenoids into the active form of Vitamin A needed for photo-reception. It turns out that blue light toxicity on the eyes or skin ruins the conversion of dietary carotenoids to retinol and this causes circadian phase delays that underpin mitochondrial diseases.
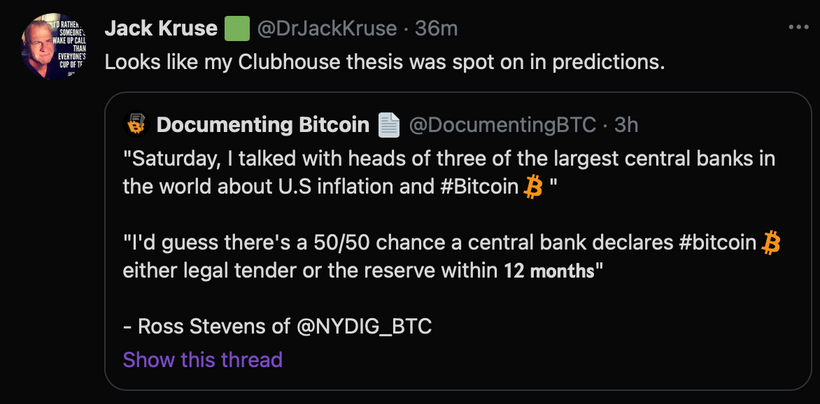
This opsin discovery is when I began to realize all I was taught about the sunlight in medical might be a fallacy. The current paradigm in ophthalmology says that UV light is really bad for the eye and skin. Well, if that was true why would Mother Nature have put a UVA opsin in the cornea and in the skin.
Yep, neuropsin has been found in the eyes, skin, and subcutaneous fat now mimicking the exact spots where melanopsin is found. Could this be why light at night leads to myopia and fat as I laid out in my Vermont 2017 video on youtube? It does appear that the sensations your eyes get from light gives us the reality we experience here on Earth.
LETS JUMP DEEPER.
The retina is the only tissue known to ignore the master clock based in the SCN, but it does keep itself on a schedule, based upon all the data we have. That made me ask the question, how does this occur?
I looked at articles where opsin were removed from animals (see above). It turned out the retinas of the mice without neuropsin lost their ability to adapt to new patterns of light and darkness. I found out that neuropsin and melatonin function must be coupled because melatonin is the hormone of darkness that is regenerated by daytime later morning UV light (9-11AM). Was neuropsin key in this recycling of melatonin? Does neuropsin also work with UV light in some way during the day?
The scientists working with these mice began by repeating their experiments with different wavelengths of light, the they found that neuropsin responds to UV-A and violet light up to 400 nm. This implied that the retina uses separate light signals based upon frequency to set its own clock and that of the body’s master clock in the SCN. The central retinal pathways controlled by melanopsin are exquisitely sensitive to light in 435nm-465nm light which is based in the blue/green part of the spectrum. 400-430 nm light is present in early AM sunlight, before UVA shows up, and it becomes very prominent at dusk when the sun slowly sets. These frequencies of light are ideally absorbed by melanopsin over several hours at dusk. Melanopsin can be thought of as an opsin that pays attention to light as it dims. Regarding neuropsin, we still don’t know precisely what kind of signals, either photonic, chemical or electrical, that neuropsin uses to set the retina’s clock. My current bet it will be found to be photoelectric because of another clue the eye has given us. Neuropsin is found in the cornea and the skin and this makes it very unique. Why would we have a UV-A light sensor for bright mid-day light frequencies in a transparent cornea? Since neuropsin works via UV-A light this is a “tell” that our modern beliefs that the cornea and lens block UV light is a pure fallacy. It also tells us that non UV-B sunlight is important to melatonin recycling.
How did we find out neuropsin was in the cornea? Researchers used a specialized “locator gene,” called a reporter gene, to figure out where neuropsin does its work. They found that, like melanopsin, it is located in neural cells that connect the retina to the brain via the central retinal pathways. From there though, we do not know precisely yet today where these particular cells go within the human brain. Using this technique they did mysteriously confirm it is present it in the cornea. This shocked many, including the eye doctors. The cornea was not thought to contain any UV pigments by ophthalmologists, since its job is to let light through to the rest of the eye. We now know this data needs updating.
MY BELIEFS ABOUT NEUROPSIN AND IT LINK TO UV-A LIGHT
I currently believe neuropsin was selected for by the KT event in eutherian mammals that made it through the last extinction event. During this time it is believed that photosynthesis was disrupted for several decades to 1000 years. This would have lowered the quantum yield of sunlight on the entire planet. A lowered quantum yield of sun light would have sharply lowered UVB light more than UVA light.
Watching the light at the sunrise at 15% angle gets best results for health. This angle is when the red light predominates. As the angle gets over 15% UVA shows up. Over 30% is when UVB light has arrived on most places on Earth.
Mammals that survived would have needed some way to get environmental signals to their pituitary gland to drive the thyroid hormone cycle needed to drive seasonal changes and to control reproduction. Without these two factors, no mammals would have survived very long, in a low quantum yield environment. In today’s world, we now see the same similarities in our environment but for different reasons. As a result, in humans, thyroid diseases and fertility changes are now at pandemic levels. I believe that UV-A light is critical in driving thyroid cycling in all modern mammals including humans. When we see massive spikes in hypothyroidism, it is a sign that there is a global lowering of the quantum yield of sunlight for some reason. 65 million years ago it was debris from an asteroid that cause blockade of specific frequencies of light. Today, it is non native EMF in the ionosphere that are destructively interfering with sunlight that falls to the Earth surface. This explains why we have a global Vitamin D3 pandemic and why we see massive spikes in hypothyroidism and infertility. Free T3 is needed for fertility (leptin) and for nerve function. Low free T3 levels also are associated with chronic pain and severe alterations in cardiac function. Free T3 levels have been studied in post heart surgery patients and been found to be an very good reliable outcome measure. Free T3 tends to trend with melatonin levels because both need UV light exposure to recycle themselves in mammals. Low free T3 levels is a sign of low UV light exposure.
Since eutherian mammals made it past this event horizon it stands to reason that this gene should be highly conserved in all mammals that survived. So what does the data say about my idea?
Within placental mammals, neuropsin is extraordinarily conserved, with percent identity relative to human protein 96% averaged over 31 species; exceeding the 95% percentile of all coding genes proteome wide! That conservation drops considerably at marsupials and monotremes (86%), is less striking at tetrapods (78%), and not especially remarkable at teleost fish (68%). This pattern suggests neuropsin acquired significant new adaptive functionality on the placental mammal stem, leading to marked resilience to fixation of any further variation for the last 65 million years. That is why I believe as I do today. My beliefs are always subject to new data.
Does this explain why the modern world faces a pandemic of hypothyroid disease?
It might. Why? How does this link to thyroid function? Most people in medicine today know about thyroid function but few can explain why hypothyroidism is now at pandemic levels. I think a global lowered quantum yield of sunlight on Earth maybe the reason. This sounds hard to fathom, because few really understand how the thyroid is controlled by light. In 2010, I came across a study that gave me some insight to build upon this idea that I wrote about in Cold Thermogenesis 4 and Cold Thermogenesis 6. Thyroid function, cold, and UV-A light exposure had to be linked in some way that biochemistry and medicine have not yet fully appreciated. The linkage seems to go all the way back to the last time the Earth faced a lowered quantum yield. The KT event was the last time Earth faced a “serious brown out from the sun”. Neuropsin evolution was likely how life back then dealt with the last brown out to survive the event. I believe neuropsin isoforms were re engineered in the early eutherian mammals and therapy dinosaurs to absorb in the blue range to overcome the loss of sun light. This helped them live in a low quantum yield world and also allowed them to uncouple their inner mitochondrial membranes to make more heat. This radically changed mitochondrial haplotypes in eutherian mammals. The UV-A neuropsin has re-evolved in mammals as the sun’s power was restored. The link of blue light neuropsin’s in bird and nocturnal mammals links to melatonin regeneration explains why life could survive in a world without full spectrum sunlight. After the asteroid impact UVB light would have been non existent on Earth because of the particulate matter in the atmosphere. UV-A light would have been present but rare. This is why I knew to look at the skin and eye surface for a possible link for a light sensor for blue light. I looked at the other animals that made it through the KT event beside eutherian mammals. They were the therapod dinosaurs. Their modern correlates are birds. Quails happen to be birds.
The 2010 study I mentioned earlier was about the quail brain. It mapped the bird version of neuropsin (NEUR1) to the paraventricular organ, a region of the diencephalon of nonmammalian vertebrates containing aminergic neuronal cell bodies (biogenic amine correlates in mammals) beneath the epithelial membrane lining the third ventricle. The Paraventricular region is where the PVN is located and controls the sympathetic nervous system and it very sensitive to the UV spectrum of light from the retina. The PVN is critical in the stress response and ROS signal from mitochondria from many different environmental signals. This peaked my interest because of what I imagined occurred globally 65 million years ago, to the quantum yield of sunlight, off the coast of Mexico. The modern vertebrate forebrain’s diencephalon also contains the thalamus, hypothalamus and posterior pituitary. This linked many areas of the mammal brain to vital functions.
WHERE DID NEUROPSIN COME FROM? A change in light.
The specific idea laid out in this paper was that incident UV-A and violet light would be sparsely present, but would be clearly able to be detected by the bird version of NEUR1 opsin in paraventricular neurons. This would allow signaling via cerebrospinal fluid to the pars tuberalis of the pituitary gland, inducing there thyroid-stimulating hormone was present. This, in turn, would induce the thyroid deiodinase DIO2 in tanycytes lining the third ventricle to affect fecundity and seasonal adaptations. The 3rd ventricle is filled with CSF, which is 99.8% water. Light changes the hydrogen bonding network in the water. Light and water are critical in forming life’s battery over the 4.5 billion year history on Earth. This interaction would lead to a photo-electric signal in the hydrogen bonding network in CSF that transforms light energy into the lattice of protons produced during a daylight long signal.

This signal induced T3 in the mediobasal hypothalamus, and ultimately could have induce gonadotropin-releasing hormone to the testis and ovaries to control seasonal reproduction. Light and metabolism have always been linked to fecundity in birds and mammals. These are critically important factors, as I laid out in the last chapter of my book, The Epi-paleo Rx. In this paper, they mentioned something rather startling to me about light frequencies and the bird opsin. The peak adsorption was observed to be at 420 nm (blue) in this opsin and it was not readily transferred to orthologs in other species. They found this out by studying genomic alignment and tuning residue formulas. This meant birds faced something rather unusual in their evolutionary past, and it changed their genome in a specific way tied to light. Their opsin responded to a predominance of blue light in the environment? Why would birds have an opsin that responded to blue and not UV light? Moreover, why would nocturnal mammals also have blue light neuropsin’s and why would humans have an opsin that responded to UV-A light?

The KT event fits this scenario quite well because of the brown out the two surviving animals were birds and small nocturnal mammals. I do not believe it is a coincidence that 420 nm is right below melanopsin’s ideal optical range in today’s modern versions of mammals. This made a lot of quantum sense to me considering what an asteroid would have done to the sun’s power and how it would have shuttered certain frequencies between the UV and blue band during daytime hours.
A commentary piece assigned to this article linked the the paraventricular organ to the hypothalamus in modern animals. I immediately thought about the paraventricular nucleus (PVN) in humans. I went digging and found that even though no exact mammalian anatomical counterpart exists completely, the Allen Brain Atlas shows mouse expression of NEUR1 there (blue range). Things began to become more clear. This had to be how light and water helped birds and mammals survive the KT event. They survived because they has strong regeneration programs despite the fact UV light was nearly absent after this event. These animals were able to navigate a low quantum yield event, by innovating an opsin in the skin and eye that responded best to a world that had buried the sun because of a bollide. This version of opsin allowed them sustain higher heteroplasmy percentages because they used the relative darkness of their environment to control regeneration. Melatonin was more important to these animals 65 million years ago than leptin was due to the decreased amount of daytime UV light. Plants are also capable of using blue green light in their photosynthetic centers too to sustain the food webs to allow for a recovery.
The hormone that controls dark = melatonin. When quantum yields are low from sunlight for any reason, sunlight would be like AM sunlight is today up to an angle of 30%, darkness would be more plentiful. There would have been equal parts of blue, green, and red with some critical UV-A light after such an event. The particulate matter would have lowered the amount of UV light considerably in the UV-B range. This environmental change of light might have optimized neuropsin protein to UV-A light.
This is how I believe neuropsin and melatonin made their alliance from an evolutionary perspective 65 million years ago in birds and eutherian mammals. This all of course, assumes that the photoperiodic reproduction hormomal control had to be directed by the light and dark mechanism under melatonin’s control. It should be no surprise that even today, melatonin levels control mitochondrial DNA heteroplasmy percentage. Leptin does not control this process. This tells me on evolutionary time scales varying light on Earth had to be intimately involved with this biologic change. This was a big clue for me why we are seeing the diseases we do today when we now live indoors under man made blue light. This gives melatonin the master control switch that deals with the percentage of heteroplasmy and mito-nuclear co-aptation that Doug Wallace has laid out for modern biology.

This melatonin/neuropsin mechanism is retained from the ancestral situation in all animals that survived the KT event. Even today, the paraventricular organ is known from teleost fish and frog as well as birds. Photoentrainment of daily circadian activity rhythms should be expected to be quite distinct in terms of opsin use from photoreceptor control of seasonal reproduction. This idea fits precisely with what we now know about the human NEUR1 transcripts most commonly recovered from testis and ovary. Want some proof for this speculation I am bring forth to you here? Google neuropsin transcript DB097202. It has a massive phylogenetic range back to amphioxus and sea urchins, past which, unfortunately nothing is known today.

SUMMARY
Knowing is not enough; we must apply the knowledge. Willing is not enough; we must do the right thing around light in the decentralized web Nature gives us on Earth or it will sculpt us in ways we not be ready for.
Nature programs in us builds endless optimism in the face of repeated failures. In this way she forces us to move to act to learn about our world. You have to open up to the world and learn optimism. In Nature, you always collect failures on your way to success.