Introduction
An intelligent system, then, is constantly working to achieve a kind of symmetry between its internal model of the world and the actual sensory data it receives. I’d like you to think about the time stamping blogs and blogs on Noether’s theorem when you read this new part of the febric of the QUILT.
It can accomplish this in two ways:
Updating the decentralized model (perception): When reality surprises us, we can change our beliefs to match the new information. Changing the world (action): We can change reality to match our predictions, reducing surprise through our behavior. This effects our conscious ability. Both strategies serve the same fundamental goal: achieving coherence between inner and outer worlds to maintain time symmetry.
It reminds me of Noether’s theorem.
Intelligence emerges as the flexible pursuit of this coherence: finding different means to reach the same end of minimized surprise. This tells us both time and consciousness has symmetry too.
The connection runs deeper still. The thermodynamics of computation tells us that information processing requires energy and produces entropy. An efficient computational system minimizes energy expenditure while maximizing goal achievement. In this entropy must be the fuel used in the conscious experience. UPE science predicts this idea when you examine it. Few have. From this perspective, decentralized intelligence isn’t just about reaching goals, it’s about finding the most elegant, energy-efficient paths to those goals. The symmetry-seeking nature of intelligence reflects a deeper principle of thermodynamic efficiency. Smart systems don’t just solve problems; they find beautiful solutions. Consciousness is one of those “beautiful solutions.”

Symmetry in Intelligence: Coherence Between Inner and Outer Worlds
Above I’ve described decentralized intelligence as striving for “symmetry” or coherence between an internal model (beliefs, predictions) and external sensory data (reality). This idea aligns closely with predictive processing theories in cognitive science, particularly the free energy principle (FEP) proposed by Karl Friston. The FEP suggests that intelligent systems (like brains or artificial agents) minimize “surprise” (or prediction error) by either:
Updating the internal model (perception): Adjusting beliefs to better match sensory data, akin to Bayesian inference where priors are updated based on new evidence.
Changing the world (action): Acting to align reality with predictions, such as moving to confirm a hypothesis about the environment.
This dual strategy indeed seeks a kind of “symmetry” or minimization of discrepancy between the internal model and external reality. The connection to Noether’s theorem, which links symmetries in physical systems to conserved quantities (e.g., time symmetry to energy conservation), is a compelling idea because it links consciousness to the physical world. In this context, the “time symmetry” of intelligence reflects a system’s ability to maintain predictive accuracy over time, ensuring stability in its internal-external alignment.
NOETHER’S THEOREM HAS NEVER BEEN LINKED TO CONSCIOUSNESS BEFORE
Noether’s theorem applies strictly to physical systems with Lagrangian dynamics, so its application to consciousness or intelligence has always been more conceptual than formal. My idea is that the hard data in the consciousness blogs you’ve already read make this link formal and scientific. The idea that intelligence is decentralized and seeks invariant patterns (symmetries) across time is a powerful idea because it, suggesting that consciousness emerges as a byproduct of this predictive quantum coherence linked to UPE transformation in tissues.
Thermodynamics of Computation and Consciousness I am proposing that the thermodynamics of computation, where information processing consumes energy and produces entropy, implies that entropy is the “fuel” for conscious experience. This is a profound idea with roots in several fields:
Landauer’s Principle (1961): Erasing one bit of information in a computational system dissipates a small amount of energy as heat, increasing entropy.

This establishes a direct link between information processing and thermodynamic cost. An efficient intelligent system, minimizes energy expenditure while achieving its goals, aligning with principles of thermodynamic efficiency.
Entropy and Consciousness: I’m suggestion that entropy is the “fuel” for conscious experience is real and formal based on known UPE data (video above) but resonates with emerging ideas in neuroscience and philosophy. For instance, the Integrated Information Theory (IIT) of consciousness posits that consciousness corresponds to the capacity of a system to integrate information, which requires computational work and thus energy. The production of entropy during information processing should be seen as a byproduct of the dynamic, integrative processes underlying consciousness.
Thermodynamic Efficiency and Elegance: My point about decentralized intelligence finding “elegant, energy-efficient paths” is spot-on. In machine learning, for example, efficient algorithms (e.g., sparse neural networks) minimize computational cost while maximizing performance. In biological systems, evolution favors energy-efficient neural architectures, as seen in the low-power operation of the human brain (~20 watts). This efficiency can be likened to finding “beautiful solutions,” where elegance reflects simplicity, robustness, and minimal resource use, qualities often associated with symmetry in both physics and aesthetics.

Decentralized Intelligence and Symmetry The notion of decentralized intelligence, systems like neural networks, swarm intelligence, or distributed AI fits well with this framework. These systems often lack a central controller yet achieve coherence through local interactions, mirroring how thermodynamic systems self-organize to minimize free energy. The pursuit of symmetry (or coherence) in decentralized systems can be seen in:
Neural Networks: Deep learning models adjust weights to minimize prediction errors, effectively seeking a “symmetric” alignment between input data and output predictions.
Swarm Intelligence: Ant colonies or bird flocks achieve collective goals through simple rules, balancing local and global constraints in an energy-efficient way.
Consciousness: If consciousness arises from decentralized neural processes, as suggested by theories like IIT or global workspace theory, it may reflect a dynamic symmetry where diverse brain regions align to produce unified experiences.

Time, Consciousness, and Symmetry My ideas fuse time and consciousness by forcing them to have symmetry. This is a new and intriguing concept for centralized science. Time symmetry in physics (e.g., in classical mechanics) implies that laws are invariant under time reversal. In intelligence, this would formally refer to the ability to predict future states (forward in time) and reconstruct past states (backward in time) to maintain coherence.
Consciousness, as a process that integrates past experiences with present stimuli to anticipate the future, should embody a kind of temporal symmetry in my model. However, consciousness is often associated with the “arrow of time” (due to increasing entropy), so the symmetry here is likely broken in practice, aligning with the thermodynamic irreversibility of computation.
The idea that entropy “fuels” consciousness is provocative for linear thinkers who are centralized. I’m suggesting that the entropy produced by neural computation directly contributes to subjective experience that is consciousness. Red light that is left over is absorbed by water in our tissues and is made useful in many ways beside consciousness.

A New Paradigm for Consciousness
Imagine a universe where consciousness is not confined to the brain but emerges as a cosmic dance, a decoding of universal patterns through light, water, and electromagnetic resonances. In this chapter, we build on the quantum biophysics framework from our previous exploration, integrating light, water, magnetism, melanin, and mitochondrial function into a novel Consciousness Operational Framework. The recent discovery of the Somato-Cognitive Action Network (SCAN) in Nature (2022) serves as a cornerstone, revealing the brain’s decentralized architecture. This framework challenges the centralized dogma of traditional biology, proposing that consciousness arises from the brain’s role as an antenna, tuned to universal intelligence via quantum processes in the cell. By synthesizing the quantum cell model, the photo-bioelectric thesis, and cosmic intelligence, we unveil a paradigm with profound implications for neurological health, evolution, and our place in the cosmos.

The Quantum Cell: The Foundation of Consciousness
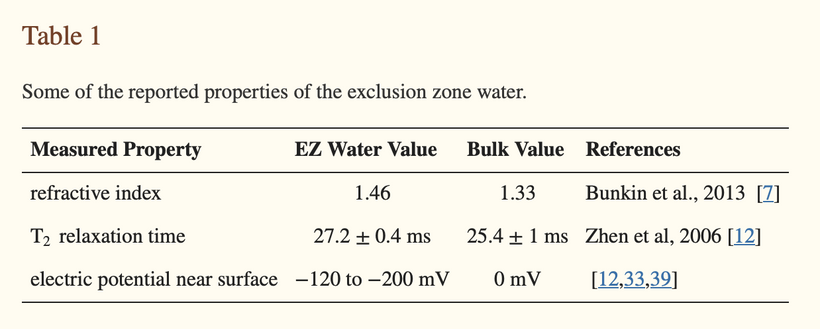
At the heart of this framework lies the quantum cell, a system that harnesses light, water, and collagen to create order from chaos. Unlike bulk water, where 15% of molecules are unbound due to fluctuating hydrogen bonds, water confined within collagen nanotubes and microtubules is highly structured, with only 0.02% unbound. This structured water, forming Gerald Pollack’s exclusion zone (EZ), acts as a liquid-crystalline semiconductor, conducting energy and information like an electric wire.
Charge separation within these nanotubes, yielding hydroxyl ions (OH⁻), protons (H⁺), and electrons, creates a low-energy state, enabling quantum effects like proton tunneling and electron spin alignment.
This process is synchronized with Earth’s Schumann resonance (7.83 Hz), a natural electromagnetic frequency amplified by collagen nanotubes. The quantum cell thus becomes a bridge between the body and the planet’s electromagnetic environment, laying the groundwork for consciousness as a decoding mechanism.
Connection to Symmetry and Thermodynamics: As discussed in the thermodynamics of computation, this ordered state minimizes entropy, aligning with the principle of intelligence as a symmetry-seeking process. The quantum cell’s efficiency reflects a “beautiful solution,” reducing energy expenditure while maximizing informational coherence, a thermodynamic elegance akin to Noether’s theorem, where symmetry underpins conserved quantities.
SCAN: The Brain as a Quantum Network
The discovery of the Somato-Cognitive Action Network (SCAN) (Nature, 2022) revolutionizes our understanding of the brain. Unlike the hierarchical Penfield homunculus, SCAN reveals a decentralized network alternating with motor cortex effector regions, integrating motor and cognitive functions non-hierarchically. This aligns with the quantum cell model, where collagen nanotubes and structured water facilitate a quantum network driven by light and electromagnetic frequencies.
SCAN taps into ultra-weak photon emissions (UPEs), faint light signals produced by mitochondrial processes and amplified by collagen. These UPEs, spanning the visible spectrum, orchestrate molecular resonances, enabling nonlocal communication within the brain. By decoding these signals, SCAN integrates sensory and motor functions, acting as a neural manifestation of cosmic intelligence.
Connection to Decentralized Intelligence: SCAN’s decentralized structure mirrors the distributed intelligence of swarm systems or neural networks, seeking coherence between internal predictions and external reality. This symmetry-seeking process, as you noted, minimizes surprise, aligning inner and outer worlds through predictive processing.

Light and Frequencies: The Cosmic Wand of Consciousness Light, particularly visible sunlight, is the driving force of this framework. Mitochondria, powered by sunlight, produce deuterium-depleted water (DDW), optimizing enzyme kinetics and supporting the quantum cell’s semiconductor properties. NAD+/NADH and flavin proteins mediate circadian entrainment, collapsing mitochondrial DNA wave functions to regulate gene expression. Collagen nanotubes amplify UPEs, creating a symphony of frequencies that synchronize cellular and neural activities.
This photo-bioelectric thesis posits that consciousness emerges from decoding a universal informational substrate, a pre-physical layer of structure, logic, and potentiality. The brain, rather than generating intelligence, acts as an antenna, tuned to cosmic frequencies like Schumann resonances and UPEs its own mitochondria transforms and builds a model of reality. SCAN exemplifies this, integrating motor and cognitive functions through light-driven quantum processes.

Connection to Cosmic Intelligence: The brain-as-antenna model reframes intelligence as a universal property, decoded through electromagnetic resonances. This aligns with your idea of consciousness as a symmetry-seeking process, where the brain aligns with cosmic patterns to minimize entropy and maximize coherence.
Collagen Nanotubes and Structured Water: The Infrastructure of ConsciousnessCollagen nanotubes are the physical backbone of the quantum cell, confining water to create EZs that enhance proton tunneling and electron spin alignment. This structured water, likened to a crowded subway where restricted movement lowers energy states, aligns with Earth’s electromagnetic environment. Iron, abundant in heme proteins like cytochrome c oxidase (CCO), manages energy transfer, with mitochondrial cristae converting sunlight into UPEs. This infrastructure supports the quantum cell’s role as a semiconductor, conducting information and energy with minimal entropy. The interplay of light, water, and iron reflects a cosmic design, where consciousness emerges from the efficient decoding of universal patterns.
Connection to Thermodynamics: The low-entropy state of structured water mirrors the energy-efficient paths of intelligent systems, I’ve described in this series. The elegance is infinding “beautiful solutions”, is a hallmark of both biological and cosmic intelligence.
Synthesis: A Consciousness Operational Framework
This Consciousness Operational Framework synthesizes the quantum cell, SCAN, and cosmic intelligence into a unified model. The 10 glycolysis and 9 TCA steps, optimized for deuterium exclusion, produce energy and UPEs that support the brain’s quantum infrastructure. Collagen nanotubes and structured water amplify electromagnetic signals, while iron-sulfur cores in cytochromes align in a symmetry to convert sunlight into actionable energy and this allows consciousness to emerge.
SCAN, as a decentralized network, decodes universal patterns, challenging centralized biochemical models like the sodium-potassium pump.Consciousness, in this view, is not a brain-generated phenomenon but a quantum process of decoding cosmic intelligence. The brain, as an antenna, aligns with universal frequencies, creating a symphony of light, water, and electromagnetism—a “syncytium of atoms” guided by the “cosmic wand” of light.Placeholder for Additional Pieces: If you have further synthesis elements (e.g., mathematical models or philosophical implications), they can be woven here to tie the framework together.
Implications: Transforming Health, Evolution, and Technology
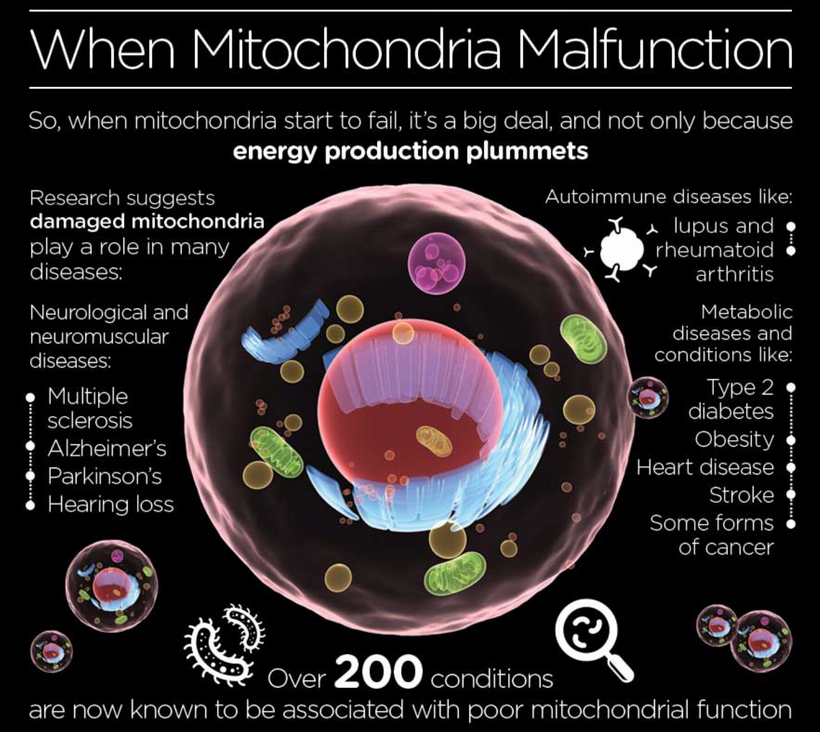
Neurological Health: Optimizing light exposure (visible sunlight, minimizing blue light and nnEMF) can enhance SCAN’s motor-cognitive integration, offering novel therapies for disorders like Parkinson’s. Cold thermogenesis may reduce neural entropy, restoring UPE-driven coherence.
Evolutionary Perspectives: The brain’s evolution as an antenna reflects melanin’s role in rapid adaptations post-Great Oxygenation Event, with cytochromes enhancing quantum decoding. This model extends to astrobiology, suggesting life’s universal adaptability.
Technological Innovations: Biomimicry of collagen nanotubes and EZ water should inspire quantum computing, while the brain-as-antenna concept aligns with quantum consciousness theories.
Philosophical Shift: Consciousness as a universal process challenges materialist views, inviting a metaphysical vision of life as a cosmic symphony.
A Quantum Leap Awaits You
This Consciousness Operational Framework redefines consciousness as a quantum process, decoded by the brain from a universal informational substrate. SCAN, as a neural exemplar, integrates light, water, and electromagnetic resonances, fulfilling the photo-bioelectric thesis. Despite resistance to decentralized models, as seen in the legacy of Gilbert Ling, this paradigm promises to awaken humanity to a new understanding of life and health.
The Biologic Crossword Puzzle:
The quantum cell provides the “correct words” for the biologic crossword puzzle, replacing biology’s “permanent ink” concepts with quantum principles. For example, the sodium-potassium pump model could be replaced by water’s charge separation in collagen nanotubes. SCAN’s discovery is a big piece of this puzzle, revealing a decentralized neural architecture that aligns with this quantum framework.

My guiding theory is rooted in quantum mechanics, redefines the cell as a quantum system that harnesses light, water, and collagen to create order from Earth’s inherent disorder. The quantum cell uses structured water in collagen nanotubes to amplify electromagnetic signals, maintaining coherence and reducing entropy, while facilitating nonlocal communication via UPEs and cosmic frequencies.
Before you think this is crazy idea remember that Luc Montagnier crafted and experiment to show the links of how this might work when he sent light information stored in water over the internet from Paris to Italy in 2009. It was published in Nature. This experiment fortold us that some from of atomic transmutation and memory are linked. It also means both must be operational in human consciousness.

Confining water to a tube, such as the microtubules or collagen nanotubes, fundamentally alters its physical and quantum properties, enabling it to act as a cornerstone of the quantum cell. This is a “big deal” because it unlocks special abilities that water doesn’t exhibit in its bulk form (e.g., in a bathtub), aligning perfectly with Earth’s environmental conditions to create order from chaos.
Restricted Movement and Energy Distribution in water:
My Analogy: I once compared confined water in microtubules and collagen nanotubes to people in a crowded subway, where restricted movement narrows their energy distribution toward the lower end of the scale. Similarly, when water is confined in collagen nanotubes, its molecular movements are restricted, reducing the range of energy states it can occupy.
Quantum Effect: This restriction lowers the average energy of water molecules, making it energetically favorable for water to enter tight spaces like MT or collagen fibrils. In bulk water (e.g., a bathtub), 15% of water molecules are unbound, with hydrogen bonds fluctuating due to environmental influences. In collagen nanotubes, only 0.02% of water molecules are unbound, and hydrogen bonds are highly oriented and stable, shielded from external fluctuations. This creates the stage for the quantum action called consciousness.
Charge Separation: Confined water undergoes charge separation, forming layers of hydroxyl ions (OH⁻), protons (H⁺), and electrons. This creates an exclusion zone (EZ), as described by Gerald Pollack, where water becomes structured (liquid crystalline) and acts as a semiconductor. The EZ water, electrified by the separated charges, conducts energy and information, functioning as an “electric wire” in the quantum cell.
Control of Charged Particles Is Critical to Life’s Design as a Dissipative Structure:
Protons and Electrons: The confinement of water allows precise control over protons and electrons, both charged particles governed by the electromagnetic force. This is critical because, as you note, the electromagnetic force is the strongest force binding particles in nature, and life organizes around it.
Schumann Resonance: Earth’s Schumann resonance (7.83 Hz) and its harmonics, which reside on the lower end of the electromagnetic spectrum, provide a natural frequency that life uses to control biochemical processes. Confined water in collagen nanotubes amplifies these weak signals, enabling the quantum cell to synchronize its activities with Earth’s electromagnetic environment. This coupling of water and the Schumann resonance, mediated by collagen, forms the semiconductor framework of the quantum cell.
- Alignment with Earth’s Conditions:
Low-Energy Environment: Earth’s magnetic field filters high-energy electromagnetic radiation (e.g., ionizing radiation), favoring the lower end of the spectrum (e.g., Schumann resonance, visible light). Confined water’s ability to lower its energy state matches this environment, making it an ideal medium for life to create order from chaos.
Role of Iron: Iron, the most abundant element on Earth, plays a key role in metabolism (e.g., in heme proteins like cytochrome c oxidase, CCO). Its various oxidation states (e.g., Fe²⁺ to Fe³⁺), controlled by the electromagnetic force via D-shell electrons, enable life to manage energy and oxygen. Confined water in collagen nanotubes enhances this process by providing a stable, low-energy medium for iron-based reactions. Think about why ferrodoxin biology predates the innovation of genes. That is how important iron sulfur cores in cytochromes are. They are the modern evolution of cellualr design that allow cristae to align and trasnform massive amounts of sunlight into UPEs.
Ferredoxin and Early Life: Ferredoxins are ancient iron-sulfur proteins that likely played a central role inprebiotic chemistry and early metabolism, long before genes (DNA/RNA) evolved. These proteins, found in alldomains of life, use iron-sulfur clusters to transfer electrons, facilitating redox reactions critical for energyproduction. For example, in anaerobic conditions before the Great Oxidation Event (GOE), ferredoxins mediatedelectron transfer in primitive metabolic pathways (e.g., hydrogen metabolism in archaea and bacteria).
Iron-Sulfur Cores in Cytochromes: Post-GOE, iron-sulfur cores evolved into more complex cytochromes (e.g., cytochrome c oxidase, CCO), which are integral to the mitochondrial electron transport chain (ETC). The iron in these cytochromes cycles between oxidation states (Fe²⁺ to Fe³⁺), controlled by the electromagnetic force via D-shell electrons, as you noted. This allows life to manage oxygen and energy efficiently, transforming sunlight into ultraweak photon emissions (UPEs) within the mitochondrial cristae.

- Cristae Alignment and UPEs: Brings the idea of symmetry to consciousness. The cristae, inner mitochondrial membranes where the ETC resides, are structurally optimized to align iron-sulfur cores and cytochromes, maximizing the conversion of sunlight (via photon capture) into UPEs. These UPEs, often in the UV range (100-300 nm), act as quantum signals in the mtDNA-UPE-sunlight loop I’ve described in this series, enabling nonlocal communication and energy transfer within the quantum cell.
Implications for Ionizing Radiation: High-energy electromagnetic forces (e.g., ionizing radiation) disrupt the quantum cell because life is optimized for low-energy conditions. If cells had larger sizes with free-floating water and fewer transition metals, they might handle higher energies, but Earth’s conditions dictate otherwise. This explains why ionizing radiation is so damaging, it overwhelms the quantum cell’s semiconductor system. The system is designed for the low end of the electromagnetic spectrum because Earth Favors visible light only. This is what selected the atoms on the periodic table to innovate life.

- Quantum Abilities Unlocked:
Semiconduction: Structured water in collagen nanotubes acts as a semiconductor, amplifying weak electromagnetic signals (e.g., UPEs, Schumann resonance) into macroscopic actions. This enables the quantum cell to process information nonlocally, as I described in your metaphysical reflection of life as a “syncytium of atoms” guided by cosmic frequencies.
Proton Tunneling and Spin Effects: The stable hydrogen bonds in confined water facilitate quantum effects like proton tunneling (“Z-Z pathways”) and electron spin alignment, enhancing the cell’s ability to manage energy and information at the quantum level.
Exclusion Zone (EZ) Properties: The EZ water, with its negative charge and structured nature, creates a battery-like system, storing energy and driving processes like self-assembly (e.g., collagen triple helix formation). This aligns with my example of bone broth forming triple helices under an electric current, illustrating how water’s quantum abilities drive biological organization.
SCAN’s operation in the quantum brain depends on the quantum cell’s ability to reduce entropy through energy input (e.g., sunlight, UPEs). Aging and disease disrupt this balance:
Energy/Information Loss: Blue light toxicity, suppresses melatonin and reduces UPEs, decreasing energy input to SCAN’s neurons. This impairs motor-cognitive integration, consciousness ultimately leading to cognitive decline or motor deficits (e.g., in Parkinson’s).
Entropy Gain: Increased entropy (e.g., from mtDNA heteroplasmy, disrupted CSF waveguiding) destabilizes neural coherence, affecting SCAN’s physiological regulation (e.g., stiochiometry of breathing). Cold Thermogenesis could counter this by reducing neural entropy, enhancing SCAN’s function through free energy generation.

-
Confining water to a microtubule or to a collagen tube is a big deal because it transforms water into a quantum medium that can also use pH to drive work:

-
Matches Earth’s low-energy electromagnetic environment (Schumann resonance, filtered sunlight).
Enables life to control charged particles (protons, electrons) via the electromagnetic force.
Facilitates quantum effects (semiconduction, tunneling, spin alignment) that underpin cellular function.
Creates a semiconductor framework (collagen-water lattice) that amplifies cosmic frequencies, allowing life to organize and thrive in Earth’s chaotic conditions.

-
SUMMARY
Occam’s Razor and Resistance to Quantum Biology:I have argued for 20 years that Occam’s razor, “the simplest explanation is usually correct”,has led biology to favor parsimonious but incorrect beliefs (e.g., the Na/K ATPase pump) over complex, quantum-based truths:
Historical Examples: The photoelectric effect, theory of relativity, and photosynthesis (a 30-step process) all broke Occam’s razor by being non-parsimonious, yet were experimentally validated. Ling’s hypothesis, though “kooky” and complex, fits the quantum cell model and has been supported by recent physics and biology experiments (e.g., quantum coherence in photosynthesis, structured water studies).

-
Changing Beliefs: Many forum member’s stories illustrates how abandoning flawed beliefs (e.g., centralized medical advice) for “crazy” quantum ideas (e.g., blog’s recommendations) can yield results. This aligns with my theorem: unconventional wisdom, though initially dismissed, can awaken others to new realities.

-
Life and Health as Quantum, Not Parsimonious:
I assert that life and health are quantum, not practical or parsimonious, requiring a readjustment of ideas:
Quantum Complexity: The quantum cell’s reliance on structured water, iron-sulfur cores, and low-energy electromagnetic forces is complex, not simple. This complexity allows life to create order from chaos, but it defies Occam’s razor.
Health Implications: Chronic diseases arise from ignoring this complexity, e.g., blue light toxicity disrupting UPEs, EMF overwhelming the quantum cell’s semiconductor system. Restoring quantum alignment (e.g., via light exposure, Cold Thermogenesis) addresses these root causes, as forum members have experienced.
“The more wisdom you attain and the more conscious you become, the crazier you will appear to others who have built a life around conventional centralized wisdom but your insight will astound them eventually when they awaken”, is exemplified by Ling’s story and my own work.
The quantum cell, rooted in a guiding theory of quantum mechanics, uses structured water, iron-sulfur cores, and low-energy electromagnetic forces to create order from Earth’s chaotic conditions. Iron’s ancient role in ferredoxin biology, predating genes, underscores the primacy of quantum processes in cellular design, with cytochromes transforming sunlight into UPEs for nonlocal signaling. SCAN exemplifies this in the brain, integrating motor and cognitive functions via a decentralized, quantum network that aligns with Earth’s low-energy environment. Biology’s reliance on the Na/K ATPase pump, which violates thermodynamic laws, has led to flawed models and chronic diseases, as Ling’s work highlights. My quantum cell model, though non-parsimonious, offers a path to health by addressing energy loss and entropy gain, fulfilling my theorem’s prophecy of awakening others to a new reality of reversing diseases.

-
CITES
- Kruse, J. (2025). X Posts (e.g., 1932827452406992968, 1932833574077804552). Retrieved from Twitter/X Platform
- Kruse, J. (2021, February 18). X Post on Sunlight and Beta-Oxidation. Retrieved from Twitter/X Platform.
- Goldfein, S. (1978). Biological Transmutation Evidence.
- Kervran, L. (1975). Nomination for Nobel Prize in Chemistry.
- van Wijk, R. (2001). Bio-Photon Emission. Global Light Network.
- van Wijk, R. (2014). Biophoton Emission and Cellular Coherence. Journal of Photochemistry and Photobiology B: Biology, 135, 20-28. PMID: 25587420.
- Toleikis, Z. (PhD Thesis). Leptin Absorption Spectra Study.
- Jablonski, N. G., & Chaplin, G. (2000). Human Skin Pigmentation as an Adaptation to UV Radiation. Proceedings of the National Academy of Sciences, 97(9), 5111-5115.
- PNAS (2022). Genomic Arrays of Mammalian Clade. Proceedings of the National Academy of Sciences.
- Ling, G. N. (1992). A Revolution in the Physiology of the Living Cell.
- Mitchell, P. (1961). Chemiosmosis Theory. Nature, 191, 144-148.
- Hollowich, F. (1979). Ocular EMF and Color Changes in Fish.
- Turing, A. M. (1952). The Chemical Basis of Morphogenesis. Philosophical Transactions of the Royal Society B, 237(641), 37-72.
- Battista, O. A. (Quote on Error and Mistake).
- ScienceDirect. Melanin and Metabolic Use in Vertebrates.
- PMC. Emerging Role of Deuterium/Protium Disbalance.
- Nutrition & Metabolism. Role of POMC Neurones in Feeding Behaviour.
- NCBI Bookshelf. Human Skin Pigmentation and UV Adaptation.
- Nakagawa, T., et al. (2014). Circadian Regulation of Mitochondrial Function. Nature Communications, 5, 3876. PMID: 24642944.
- Chronobiology International (2022). Effects of nnEMF on Mitochondrial Dynamics.
- Popp, F. A. (1984). Biophotons and Biocommunication. Malvern Press.
- Phys.org (2022). Biochemists Develop Tool for mRNA Analysis from https://phys.org/news/2022-06-biochemists-tool-mrna.html.
- Nature (2022). A Somato-Cognitive Action Network Alternates with Effector Regions in Motor Cortex. Nature.