February 2019 Webinar – ALS and 5G
Motor neuron disease (MND) describes a group of neurological disorders characterized by the selective loss of motor neurons. Amyotrophic lateral sclerosis (ALS), the most common subtype of MND, is characterized by a progressive degeneration of both upper and lower motor neurons, resulting in muscle atrophy, gradual paralysis, and death, usually resulting from respiratory failure. ALS has a worldwide prevalence of 4–6 in 100,000, with differences noted between populations.
ALS clinically overlaps with frontotemporal dementia (FTD). ALS seems linked to the cervical spinal cord barrier (CSCB) and FTD is tied to defects seen more with the blood brain barrier in the skull. FTD is a common cause of dementia in adults under the age of 65 that is characterized by neurodegeneration of the frontal and temporal cortex. Around 50% of ALS patients display evidence of frontal and temporal lobe dysfunction on detailed neuropsychological testing and up to 5–10% of ALS cases present with clinically diagnosed FTD. Many people do not know about this linkage and I think this gives the Black swan some insight to what really maybe causing the defect. Steve Gleason, a former special teams ace with the New Orleans Saints has ALS, and Hall of Fame TE, John Mackey died of FTD many years ago after multiple concussions in his career. The link to trauma and continued electromagnetic damage to the enzymes and second messengers that control the resolution of the inflammatory and regeneration response might be the missing piece to this puzzle. If I am right, we should see the incidence and prevalence of ALS rise primarly due to the chronic electromagnetic pollution that the modern world generates.
What are you saying Jack?
Could ALS be due to an “electromagnetic defect” in the control levers of the process keeping the injury repair pro-inflamamtory for too long? Might the EMF stimulus in the environment control this energy phase transition thermodynamically? It is now well known that environmental EMF’s are capable of blocking the phase transition of the inflammatory response to the anti-inflammatory pathways via calcium ion resonance changes.
Could this be why ALS is a mystery to medicine? Does this mimic the story of the blind men and the elephant?
Is this why mitochondrial dysfunction has emerged as a common, early phenomenon in ALS literature? The appearance of deficits in oxidative phosphorylation, calcium buffering and mitochondrial transport prior to the onset of disease symptoms in vivo in disease models suggests an important role for loss of mitochondrial integrity in the etiology of ALS.
I currently believe the stimulus for most cases appears to be a combination of mechanical trauma exacerbated by light stress from parts of the electromagnetic spectrum humans are not adapted to yet.
This preferentially destorys the motor neurons in the cervical cord which leads to Wallerian degeneration because the chronic stressor is never removed because no has thought about the combination effect on our colony of mitochondria.
Motor neurons have more mitochondrial density than the sensory neurons. Anatomically, their main cell bodies are also further from the motor neuron soma making repairs more tedious and energy costly from a circadian standpoint. The dorsal root ganglion (DRG) is very close to the sensory afferents in humans and it might be why ALS spares them.

It appears ALS is now being linked to electromagnetic pollution from the epidemiologic literature.
New analysis of data from more than 58,000 men and 6,500 women suggests it is the latter. Dr. Roel Vermeulen, at Utrecht University in the Netherlands, and his team found that people whose jobs exposed them to high levels of very low frequency magnetic fields were twice as likely to develop ALS as people who have never had this kind of occupational exposure.
Jobs with relatively high levels of extremely low frequency electromagnetic fields include electric line installers, athletes, welders, sewing-machine operators, and aircraft pilots and first responders. These are essentially jobs where all workers are placed in close proximity to appliances that use a lot of electricity on a chronic basis. There are many more jobs that have this risk today in a 5G world and this is why I expect the numbers to rise for ALS.
Highlights of this disease:
•
Mitochondrial dysfunction is one of the earliest pathophysiological events in amyotrophic lateral sclerosis (ALS).
•
ALS-associated mutant proteins accumulate in mitochondria and cause mitochondrial damage.
•
ALS-associated mitochondrial dysfunction occurs at multiple levels in the nervous system.
•
ALS affects mitochondrial respiration and ATP production, calcium handling, dynamics, and apoptotic signalling of cytochrome 4 to lead to excessive destruction of motor neurons.
In the PNS, motor and sensory neurons have particularly long axons. But the primary synpse is different in each system in humans. The sensory side makes its initial synpase early compared to the motor neuron. This means the motor axon is more at risk for energy deficits. The mitochondria-associated membranes, therefore, represent extreme examples of how important the maintenance of endoplasmic reticulum (ER) and mitochondrial functions can be to sustain high axonal metabolic demand.
MOTOR NEURONS HAVE MORE MITOCHONDRIA AND MORE SYNAPSES = need more ATP or solar redox with injury
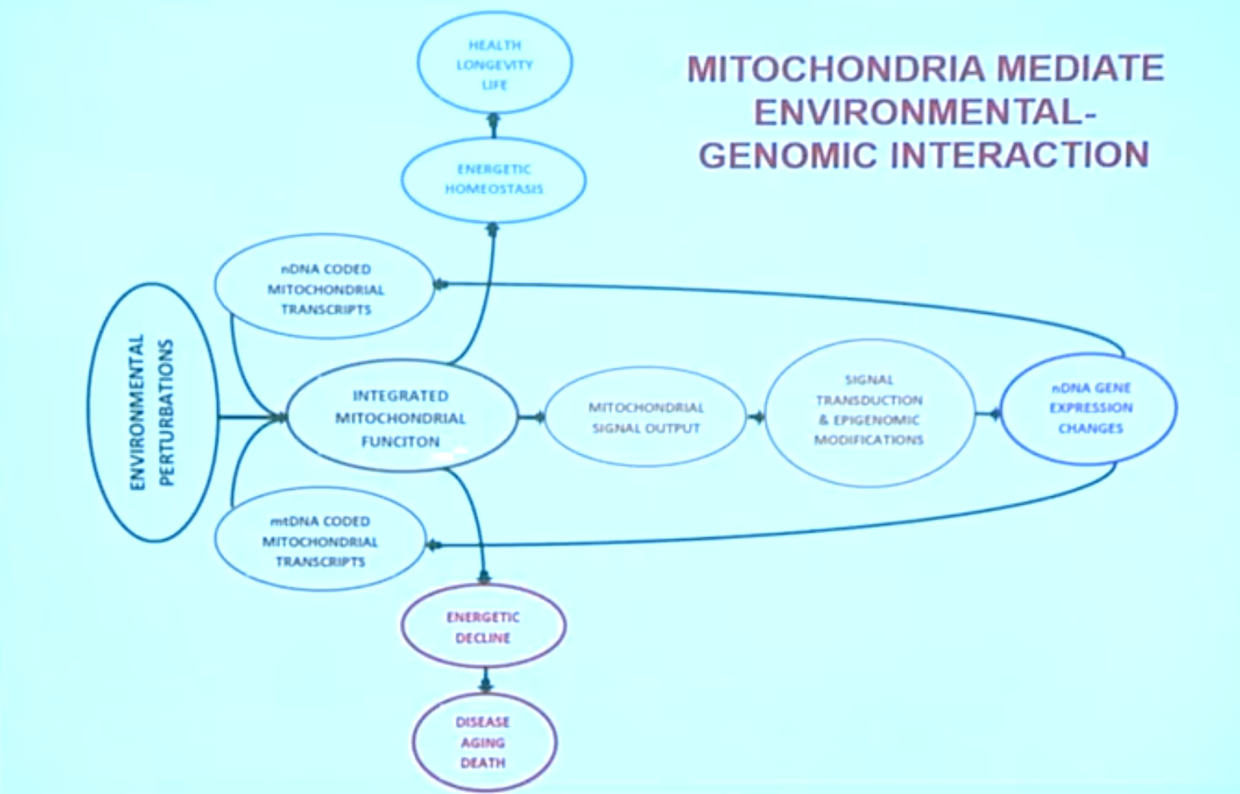
Recent progress in the understanding of neurodegenerative diseases revealed that multiple molecular mechanisms contribute to pathological changes in neurons. A large fraction of these alterations can be linked to dysfunction in the endoplasmic reticulum (ER) and mitochondria, affecting metabolism and secretion of lipids and proteins, calcium homeostasis, and energy production.
Remarkably, these organelles are interacting with each other at specialized domains on the ER called mitochondria-associated membranes (MAMs). These membrane structures rely on the interaction of several complexes of proteins localized either at the mitochondria or at the ER interface and serve as an exchange platform of calcium, metabolites, and lipids, which are critical for the function of both organelles. In addition, recent evidence indicates that MAMs also play a role in the control of mitochondria dynamics and autophagy. MAMs thus start to emerge as a key element connecting many changes observed in all neurodegenerative diseases.
Neuronal function relies on synaptic transmission, which is based on the propagation of action potentials along axons and neurotransmitter release. As the majority of biosynthetic pathways take place in the neuron soma, axons and distal synaptic contacts need efficient axonal transport for the supply of organelles and vesicles. Axonal transport is driven by motor proteins, which consume substantial amounts of energy in the form of ATP or light energy in the exclusion zone.
Sensory neurons and motoneurons have axons up to 1 meter in length. Their extreme dendrite/cell-body/axon polarization and their large soma make these neurons highly demanding in energy to function properly.
It was estimated that the anterograde transport of one vesicle along the 1 m long axon of a human motoneuron requires approximately 1.25 × 10^8 adenosine triphosphate (ATP) molecules. This is an astronomical number and tells how important mitochondrial redox is in ALS.
High metabolic demand requires a tight coordination between the production of nitric oxide (NO), ATP, melanopsin dysfunction, protein secretion, organelle biogenesis, and degradation processes that avoid accumulation of defective components to coordinate injury, repair, and regeneration sequences properly. It requires an intact circadian mechanism in the neurons and the circulation to clear the blood of small moleculare weight proteins that can lead to damage. Trauma to mitochondria is known to liberate CpG Islands which alter methylation patterns in damaged tissues.
These ideas imply that long axons are therefore particularly vulnerable to conditions of suboptimal energy supply. The axons of the cortisospinal tract are quite long. The sensory afferents are not that long because they synapse early in the CNS.
The axonal compartment often degenerates first in diseases affecting long-projection neurons, such as amyotrophic lateral sclerosis (ALS) and hereditary motor and sensory neuropathies (HMSNs) also known as Charcot–Marie–Tooth diseases (CMTs)
This leads me to believe that ALS always begins with a stressor stimulus of some type that become chronic by another mechanism to prolong the normal healing in the nerve cells to destroy regeneration of neuron function by severely altering ATP and EZ function in the spinal cord.
• Sensory neurons are situated in the dorsal root ganglion of the spinal nerve, whereas motor neurons are situated in the ventral root ganglion of the spinal cord.
• Sensory neurons follow afferent pathways while motor neurons follow efferent pathways.
• An average adult has about 10 million sensory neurons and half a million of motor neurons.
• Motor neurons are multipolar, and sensory neurons are unipolar = more synapses = more mitochondria = more energy requirements
• Sensory neuron has only afferent fiber and many proximal synapses on the soma, whereas motor neuron has efferent fibers with many distal synapses. This is an energy drain on the motor neurons in a chronic trauma situation.
• Motor neurons do not generate commands to communicate with muscles, but receive from sensory neurons.
KRUSE SPECULATIONS MADE IN THE WEBINAR:
Could trauma cause a chronic colony of mitochondrial failure in the motor neurons of ALS patients?
Can that trauma be induced many ways?
CAN LIGHT CAUSE THE TRAUMA?
CAN LIGHT PROLONG THE RECOVERY?
CAN LIGHT RUIN THE REGENERATION SWITCH?
Can the primary injury be a small incident or accident whose healing goes awry?
Can a job, like an NFL player, or athlete, a pilot, a worker for a utility company, or a smart meter on a house be all it takes to start this process?
Could the trauma be induced by the aberrant use of the electromagnetic spectrum of light?
THE ANSWER IS YES TOO ALL of them, in my opinion.
Mitochondrial ATP production depends on calcium concentration, which is controlled by the ER and that switch is calcium Calmodulin protein.
THE LINK TO TRAUMA
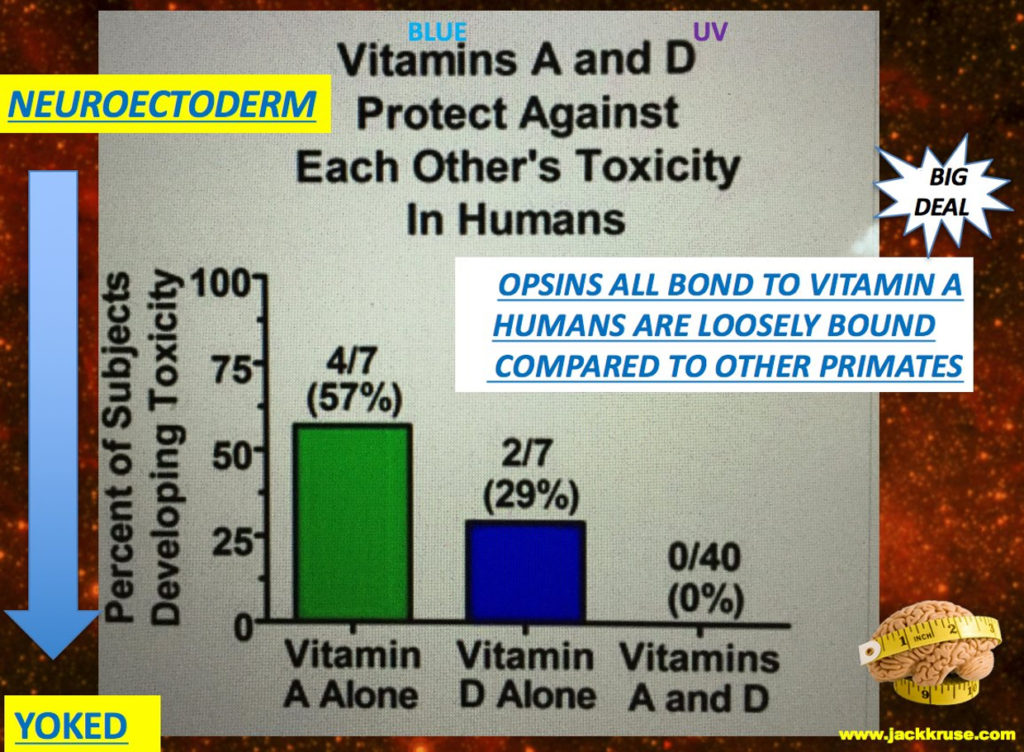
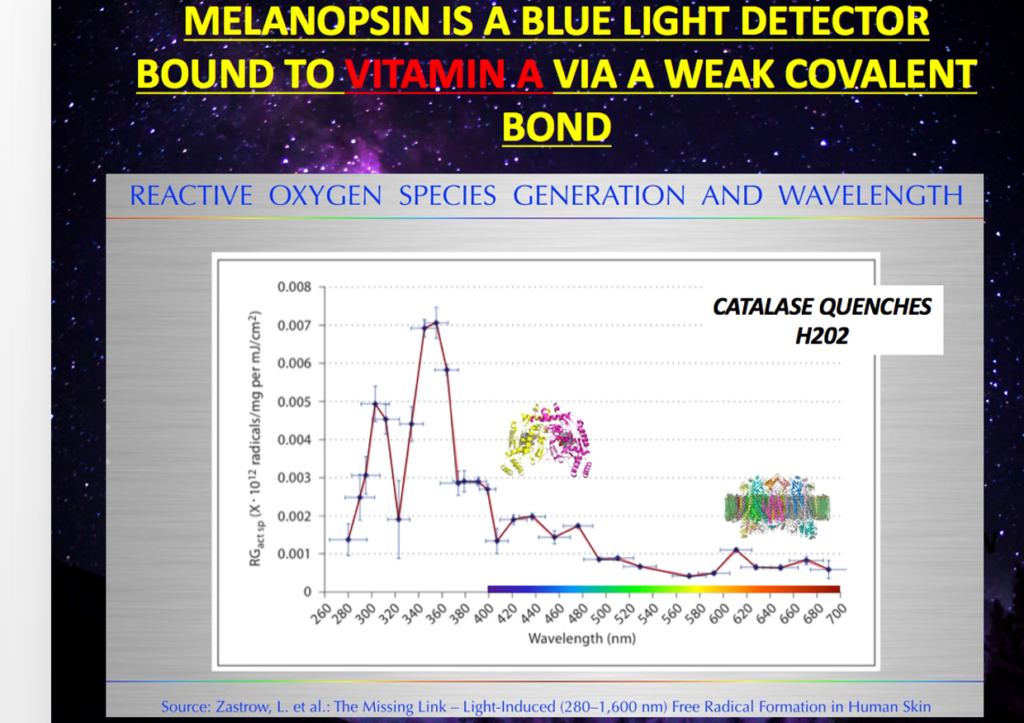
Injury to the cervical neuroforaminal stenosis spinal nerve is capable of interupting the piezoelectric and flexoelectric qualities of the collagen in the anterior and posterior longitudinal ligaments. This in turn, can electrostatically make the cervical blood brain barrier (BBB) more permeable than it should be. This leads to melanopsin dysfunction and alterations in Vitamin D and A in the blood plasma. This is a clue that the RBC’s circadian cycle is off too leaving more CPG Islands in the blood to cause more melanopsin damage.
Having a lowered Vitamin D and A level makes this mechanism even more likely. Melanopsin dysfunction is linked to lowering both Vitamin A and D via complex biophysical mechanisms. This allows both good things, for regeneration from the initial trauma to get in to stimulate healing. It also allows bad things, like CPG Islands and toxins (deuterium and proteins) to gain entrance the spinal cord to affect the metabolism of the motor neuron cells. Trauma can open the BBB of the head or the BSCB of the cervical spine to allow more DHA to enter for regeneration and repair but it could also let small proteins into the mitochondria of neurons and glial cells that lead to ALS.
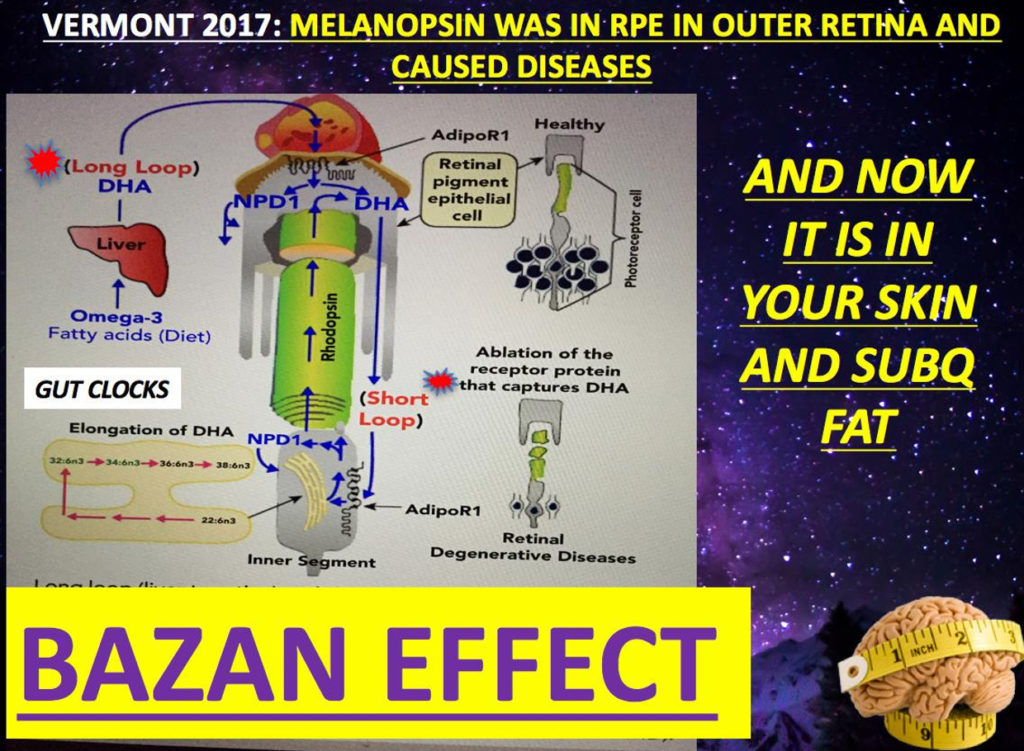
Can peripheral nerve injury alters the blood–spinal cord barrier (BSCB) and change its functional and molecular integrity through a selective inflammatory pathway? We saw this was true in the eye when the Bazan effect short and long loops where affected. Could the same type of mechanism be operational in the spinal cord? I think so.
Peripheral nerve lesion triggers alterations in the spinal microenvironment that contribute to the pathogenesis of neuropathic pain. While neurons and glia have been implicated in these functional changes, it remains largely underexplored whether the blood–spinal cord barrier (BSCB) is also involved. The BSCB is an important component in the CNS/PNS homeostasis, and compromised BSCB has been associated with different pathologies affecting the spinal cord.
In cite one below, they demonstrated that a remote injury on the peripheral nerve in rats triggered a leakage of the BSCB, which was independent of spinal microglial activation. The increase of BSCB permeability to different size tracers, such as Evans Blue and sodium fluorescein, was restricted to the lumbar spinal cord and prominent for at least 4 weeks after injury.
The spinal inflammatory reaction triggered by nerve injury was a key player in modulating BSCB permeability. The researchers identified MCP-1 as an endogenous trigger for the BSCB leakage. MCP-1 is a cytokine called monocyte chemoattractant protein 1 and also known as CCL2 in the literature.
BSCB permeability can also be impaired by circulating IL-1 in the plasma. This occurs in tisue healing in any trauma. In contrast, antiinflammatory cytokines TGF-1 and IL-10 were able to shut down the openings of the BSCB following nerve injury.
Peripheral nerve injury from many types of stimuli cause a decrease in tight junction and caveolae-associated proteins.
Interestingly, ZO-1 and occludin, but not caveolin-1, were rescued by TGF-1. Furthermore, their data provide direct evidence that disrupted BSCB following nerve injury contributed to the influx of inflammatory mediators and the recruitment of spinal blood borne monocytes/macrophages, which played a major role in the development of neuropathic pain.
Is this how whiplash causes pain too? Is this how chronic pain syndromes develop too? Peripheral neuropathy? I think so.
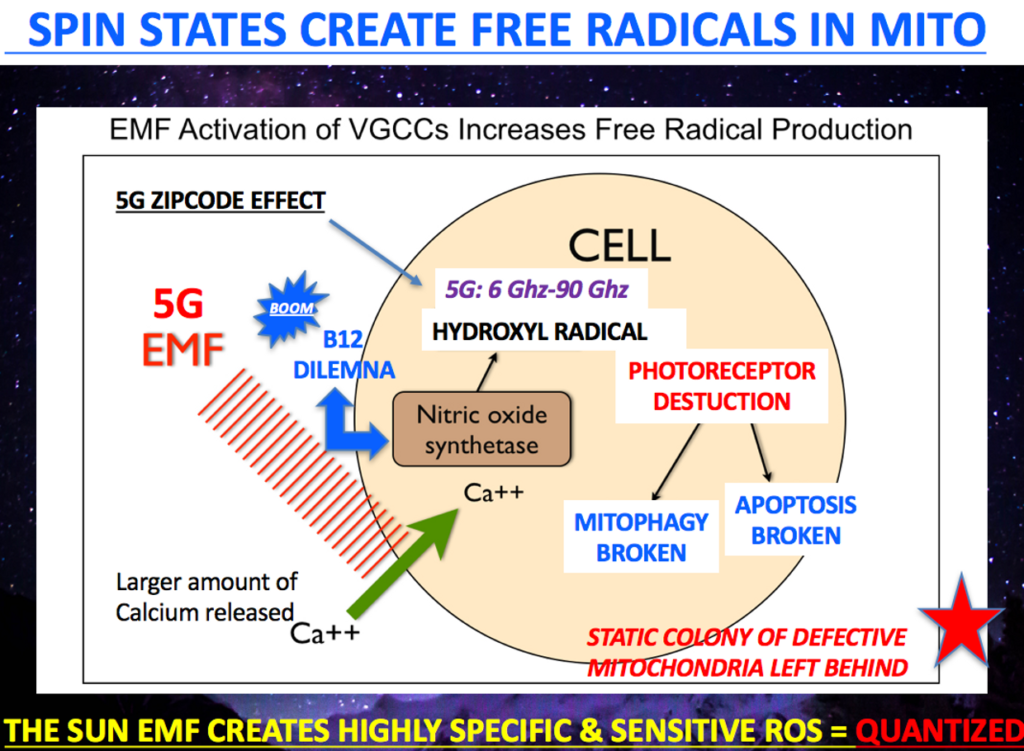
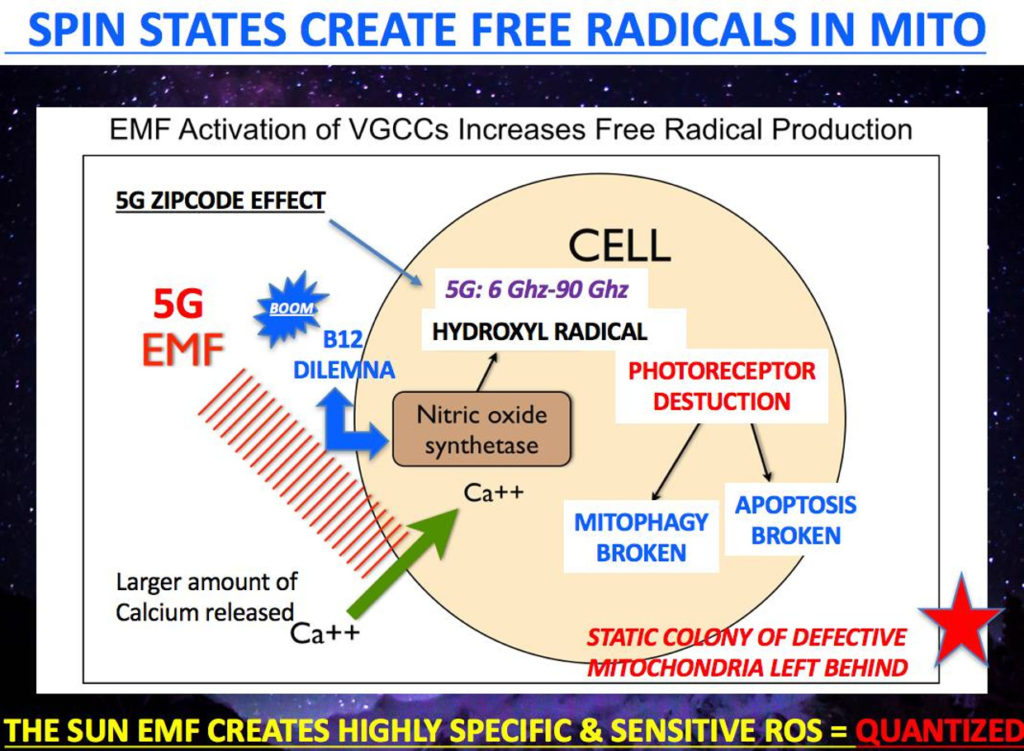
Is it possible these could be the initial steps that lead to devastating diseases like ALS if the trauma is chronic? It was found in 2013 that nnEMF releases MCP-1. It might not be a specualtion based on the data published already.
Recently, several reports have evaluated the effects of extremely low frequency electromagnetic fields (EMFs) on tissue repair. The problem with these reports is the recipe of the mechanism of action remains muddy because of poor methodology of the studies.
In particular, the data analysis supports an anti-inflammatory effect of EMFs by the modulation of cytokine profiles that drive the transition from a chronic pro-inflammatory state to an anti-inflammatory state of the healing process.
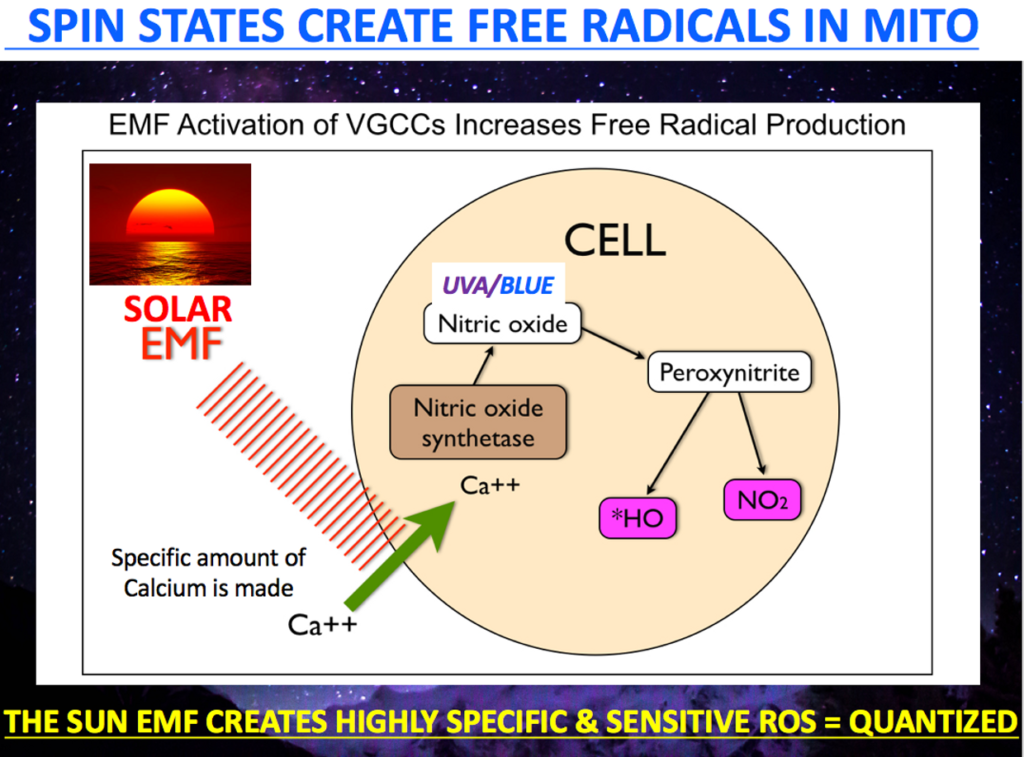
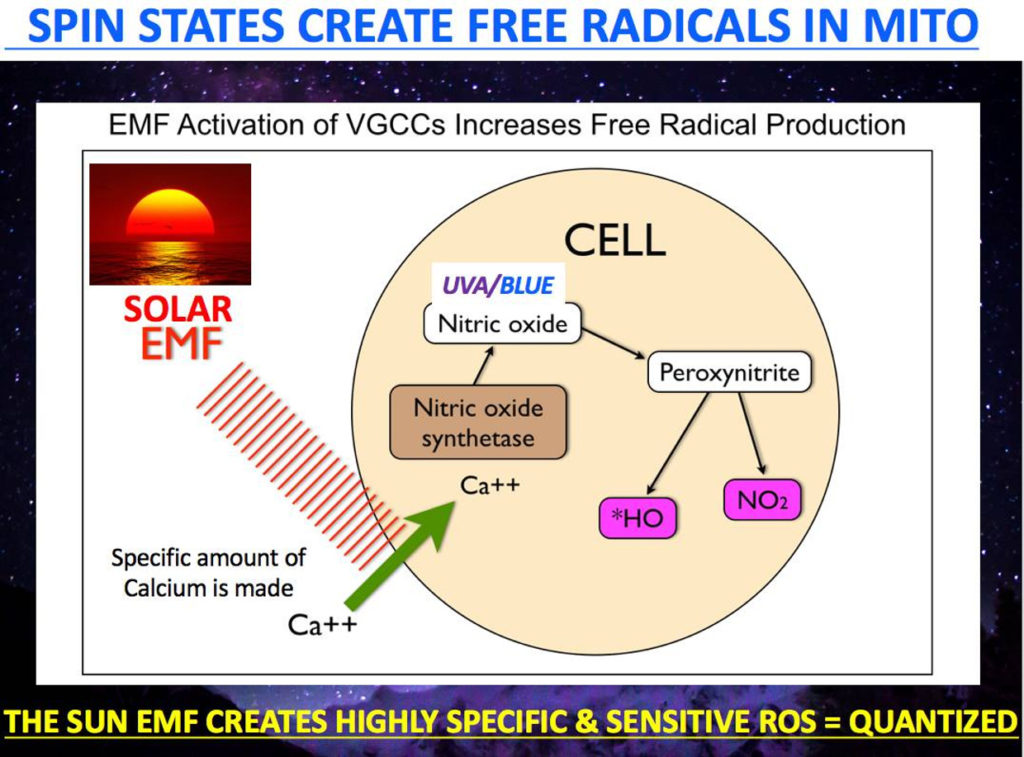
In particular, electromagnetic fields act on tissues by using light waves and calcium signaling to increasing ROS, NO and pro-inflammatory cytokines production in macrophages and following this can contribute to the establishment of “a switch” toward the resolution of the inflammatory response, and thus wound healing.
Accordingly, EMFs are capable of inducing anti-inflammatory cytokines and they contribute to the down-regulation of pro-inflammatory ones. These events can be usurped by alien frequencies if introduced into the process of healing. This ruins the cooridination of the delicate electromagnetic process.

Sunlight, nature’s key EMF induces nitric oxide production during an injury. This can be explained by the increase in the bioavailability of nitric oxide (NO) induced by exposure to EMFs in celltypes involved in the reparative process. NO shuts off CCO and this stimulus get rid of bad matrix engines. NO also shunts oxygen to better functioning mitochondria. This clears damaged tissues. I do not think medicine realizes how vital this function is in ALS. Indeed, it has been reported that EMFs activate the calcium calmodulin switch (CaM) which leads to increased activity of eNOS production and bioavailable NO in the microcirculation. This same switch is found in blood vessels where melanopsin resides!!!

At this microcirculation level, the NO is able to activate both guanylate cyclase (sGC) and adenylate cyclase (sAC) properly in the blood. This also implies getting outside and your skin in the game after a trauma induced event may be a wise thing to do since it appears light activation of wound healing is a critical step. It also helps us understand why so many concussed players are light sensitive. They have induced MELANOPSIN dysfunction and blue light exacerbates the issue.

This is why surgeons have found for years that wounds made earlier in the day heal much better than wounds created at night under fake light conditions. In fact, this is why surgeons do elective surgeries beginning so early in the AM.
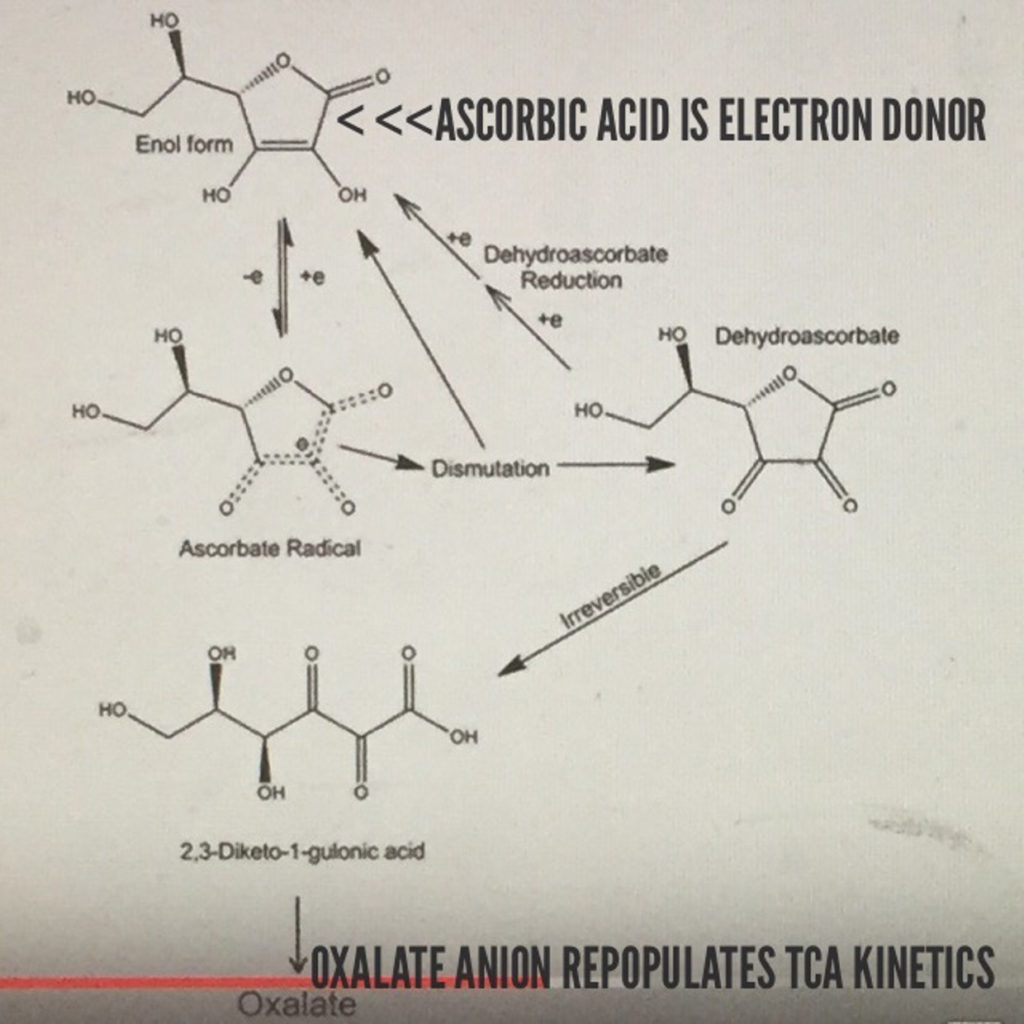
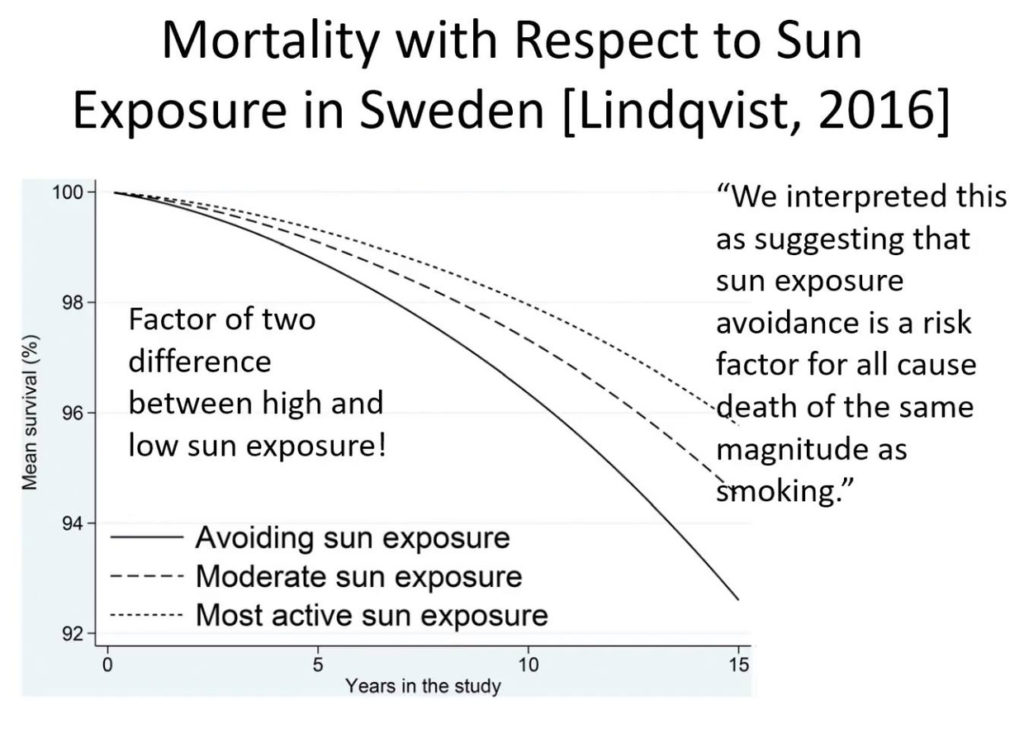
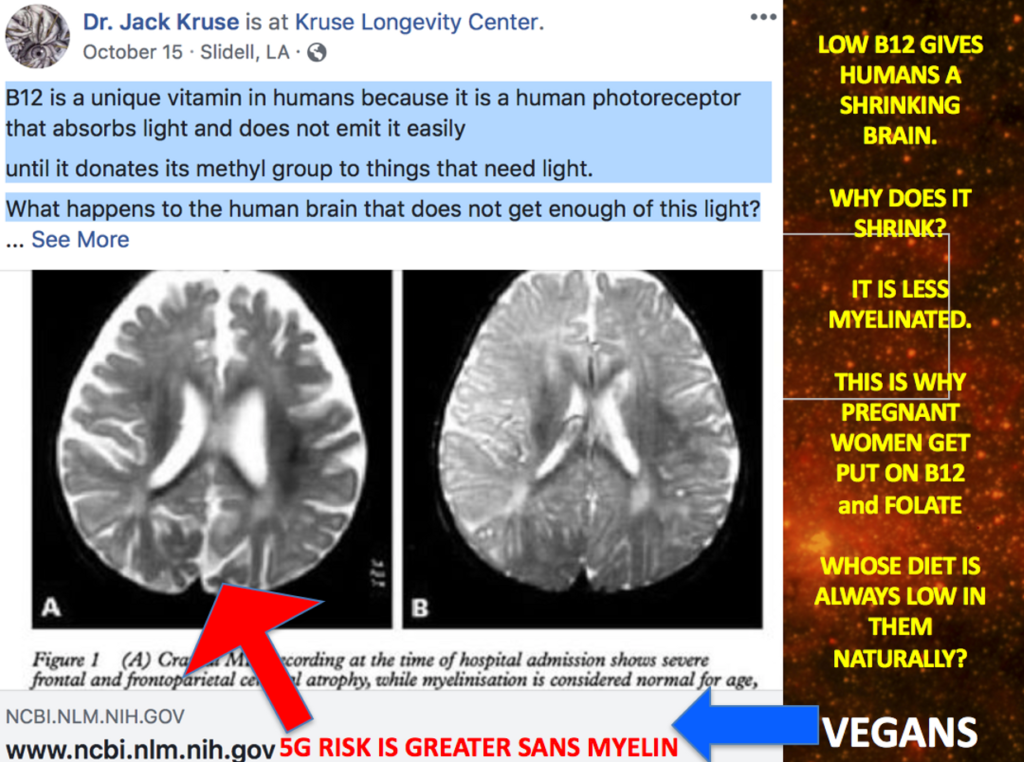
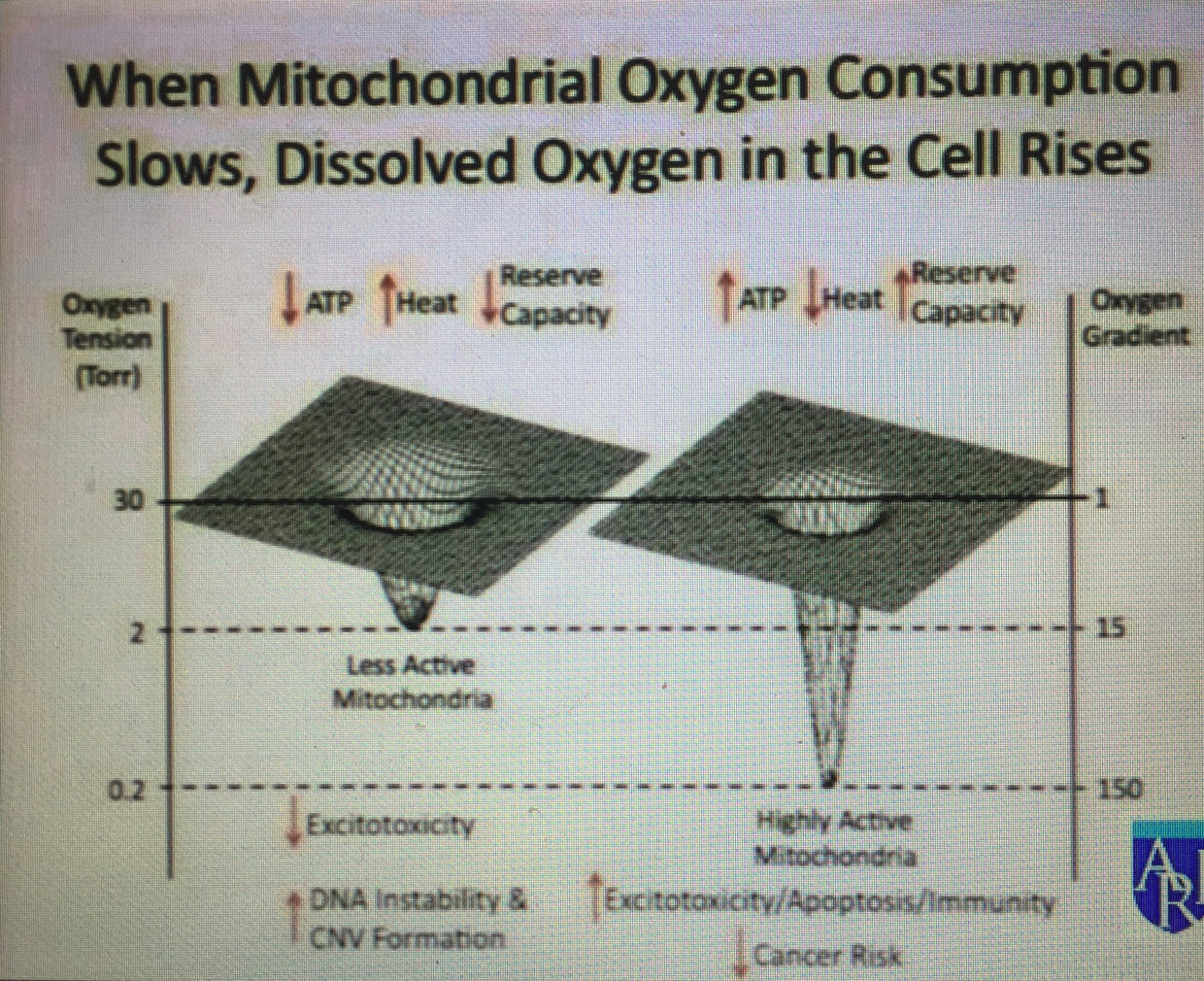
Might this mean that we should expect more ALS in people who work 3rd shifts or people who rarely go out in the sun who have low Vitamin D and A levels in their blood? I believe so. I also think these people will have very low levels of ascorbate in their plasma too to create the neurohormones needed for wound repair. When there is mitochondrial damage cells increase their oxygen levels and this GENERATES excessive ROS which leads to damage. If the trauma is chronic the ROS never resolves = apoptosis of the motor neuron.
SUMMARY:
Motor neurons have more mitochondria than sensory neurons so they are more sensitive to repetive trauma.
Defective mitochondrial respiration disrupts ATP production quickest and shoudl effect neurons with higher mitochondrial density. Is this why motor neurons are more effected in disruption of the spinal cords blood barrier to cause melanopsin damage to the vessels there?
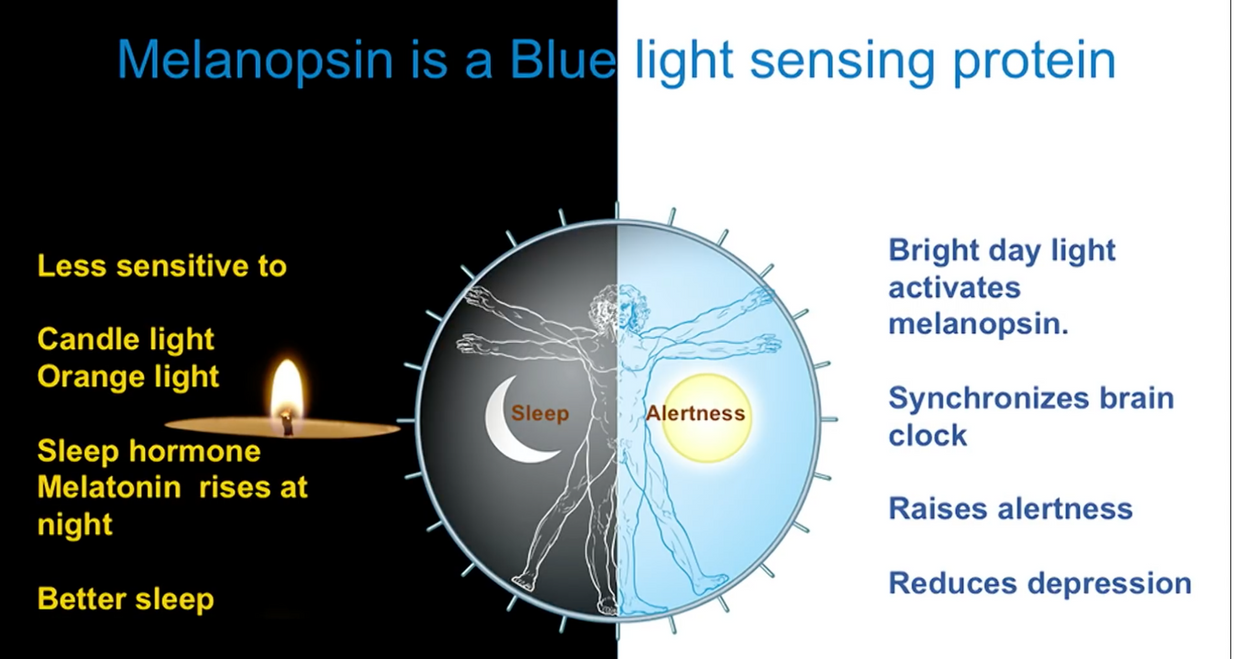
Blue light is known to lower beta oxidation and lower ATP production. Blue light also is known to cause melanopsin dysfucntion when the blue light stimulus is chronic. Melanopsin is now known to be in blood vessels since 2014.
Reductions in cellular respiration and ATP production are well documented in ALS. In post-mortem spinal cord of sporadic ALS patients the activity of all ETC complexes, complex I, II, III, and IV was found to be reduced. In addition, the activity of complexes I and IV were reported to be impaired in skeletal muscle while complex I activity and ATP levels were reduced in lymphocytes of sporadic ALS patients.
Counterintuitively, in fibroblasts obtained from skin biopsies of sporadic ALS patients the mitochondrial membrane potential (MMP) was increased compared to healthy controls. Fibroblast repair occurs under the direction of red light which makes up 42% of sunlight. There is no red light in indoor light. I think the leaky MMP might be theresponse of a patient not having their skin in the game of nature enough when they suffer chronic repetitive injuries to the head and neck from trauma or light stress. I believe ALS is a neural trauma that needs our skin in the game of sunlight quickly. I think the motor neuron needs to off switch of sunlight to repair the colony defect in the matrix of motor neurons and cannot get enough because the environment is so toxic due to electromagnetic pollution destory normal wound healing.
CITES:
1. Stefania Echeverry,1 Xiang Qun Shi,1 Serge Rivest,2 and Ji Zhang1 1 The Alan Edwards Centre for Research on Pain, McGill University, Montreal, Quebec H3A 2B2, Canada, and 2 Centre de Recherche du Centre Hospitalier de l’Universite´ Laval, Quebec, Quebec G1V 4G2, Canada
2. The Journal of Neuroscience, July 27, 2011 • 31(30):10819 –10828
5. October 2016, Townsend Newsletter — Hypothesis: Osseous Spinal Injury & Reinjury As A Risk Factor, Biomarker and Etiological Factor in Sporadic ALS
6. June 2016, Townsend Newsletter – A Glimmer of Hope About ALS Causation.
7. https://www.sciencedirect.com/science/article/pii/S030439401730544X
8. Hayashi, T., Rizzuto, R., Hajnoczky, G. & Su, T. P. MAM: more than just a housekeeper. Trends Cell. Biol. 19, 81–88 (2009).